Summary
The human gut is colonized by a diverse microbial community collectively referred to as the gut microbiota. A healthy gut microbiota provides protection against intestinal colonisation by pathogens, however disruption of the gut microbiota can weaken colonisation resistance (e.g. following exposure to antibiotics), increasing the host’s susceptibility to enteric infections. The aim of our research is to develop novel microbiome therapeutics to restore microbiota-mediated colonisation resistance and decolonise multidrug-resistant pathogens from the intestine.
Antimicrobial resistance is a serious threat to human health, resulting in treatment failures, infection relapses, longer hospitalizations, and poor clinical outcomes. The intestine is the primary colonisation site for multidrug-resistant organisms (MDROs) and serves as a reservoir for MDROs that are responsible for invasive infections (e.g. sepsis and recurrent urinary tract infections). Antibiotic treatment makes the host more susceptible to intestinal colonisation with pathogens such as carbapenem-resistant Enterobacteriaceae (CRE), vancomycin-resistant Enterococcus (VRE), and other multidrug-resistant pathogens that we are investigating in our lab. The goal of our research is to determine the mechanism(s) by which gut microbiota-mediated colonisation resistance protects the host against intestinal MDRO colonisation, with the aim to develop a new microbiome therapeutic for MDRO intestinal decolonisation. We are particularly interested in gut microbiota-mediated nutrient competition and metabolite inhibition.
Our lab uses a variety of complementary approaches to study gut colonisation resistance, including artificial gut models (aka “chemostat” or “Robogut” models), batch culture experiments, and patient faecal samples. Samples are analyzed using both culture-dependent and culture-independent techniques, including a variety of “omic” techniques (e.g. 16S rRNA gene sequencing, 1H-NMR spectroscopy, and mass spectrometry).
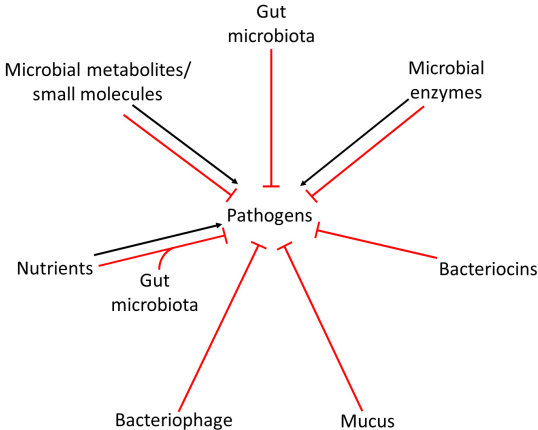
Summary of different mechanisms of gut microbiota-mediated colonisation resistance that can be used to develop new microbiome therapeutics.

Artificial gut ("chemostat") model used to study the gut microbiota.
Selected Publications
Journal Articles
Mullish BH, McDonald JAK, Pechlivanis A, et al., 2019, Microbial bile salt hydrolases mediate the efficacy of faecal microbiota transplant in the treatment of recurrent Clostridioides difficile infection, Gut, Vol:68, ISSN:0017-5749, Pages:1791-1800
Mullish BH, Ghani R, McDonald J, et al., 2019, Faecal microbiota transplant for eradication of multidrug-resistant Enterobacteriaceae: a lesson in applying best practice? Re: 'A five-day course of oral antibiotics followed by faecal transplantation to eradicate carriage of multidrug-resistant Enterobacteriaceae: A Randomized Clinical Trial', Clinical Microbiology and Infection, Vol:25, ISSN:1198-743X, Pages:912-913
McDonald JAK, Mullish BH, Pechlivanis A, et al., 2018, Inhibiting growth of clostridioides difficile by restoring valerate, produced by the intestinal microbiota, Gastroenterology, Vol:155, ISSN:0016-5085, Pages:1495-1507.e15
Mullish BH, Pechlivanis A, Barker GF, et al., 2018, Functional microbiomics: evaluation of gut microbiota-bile acid metabolism interactions in health and disease, Methods, Vol:149, ISSN:1046-2023, Pages:49-58
Mullish BH, Osborne LS, Marchesi JR, et al., 2018, The implementation of omics technologies in cancer microbiome research, Ecancermedicalscience, Vol:12, ISSN:1754-6605, Pages:1-11
Yen S, McDonald JAK, Schroeter K, et al., 2015, Metabolomic Analysis of Human Fecal Microbiota: A Comparison of Feces-Derived Communities and Defined Mixed Communities, Journal of Proteome Research, Vol:14, ISSN:1535-3893, Pages:1472-1482
McDonald JAK, Fuentes S, Schroeter K, et al., 2015, Simulating distal gut mucosal and luminal communities using packed-column biofilm reactors and an in vitro chemostat model, Journal of Microbiological Methods, Vol:108, ISSN:0167-7012, Pages:36-44
McDonald JAK, Schroeter K, Fuentes S, et al., 2013, Evaluation of microbial community reproducibility, stability and composition in a human distal gut chemostat model, Journal of Microbiological Methods, Vol:95, ISSN:0167-7012, Pages:167-174

