Drug-resistant infections
 Antimicrobial resistance (AMR) is the term used to describe the ability of some bugs to remain unaffected by certain drugs.
Antimicrobial resistance (AMR) is the term used to describe the ability of some bugs to remain unaffected by certain drugs.
'Antimicrobials' is the name given to a group of medicines which treat infections caused by microorganisms such as bacteria (for example, E. coli), parasites (for example, malaria), viruses (for example, influenza), and fungi (for example, Candida).
Antimicrobial resistance describes the changes in bacteria, parasites, viruses, and fungi which enable them to withstand treatment by antimicrobial drugs, meaning that the drugs no longer work, or work less effectively. There is a common misconception that it is the body that becomes resistant to antibiotics, but this is not the case. It is the bacteria, which cause the illness, that become resistant. You may have heard of some resistant bacteria, so-called 'superbugs' such as MRSA (Methicillin Resistant Staphylococcus aureus) in the media.
Most people will be familiar with antibiotics such as penicillin. The discovery of penicillin by Alexander Fleming - an alumnus of Imperial College - in 1928, its mass production, and widespread use during World War II and beyond have saved countless lives that would otherwise have been lost due to infection. Today, antimicrobials continue to be vital in the treatment of disease and are fundamental to the medical practices we take for granted, from chemotherapy to hip replacements.
Antibiotic Research UK
ARUK is a UK charity dedicated to serving patients with affected by AMR and they are determined to offer as much support as we can to those affected. Find out more about how they are supporting people who have been either been diagnosed with an antibiotic-resistant infection themselves, or who have cared for someone who has.
AMR
What causes drug resistant infections/antimicrobial resistance?
 Drug resistance is a natural process caused by bacteria, parasites, viruses or fungi developing to survive in their environment. In the fight against these bugs, some of them will eventually learn to adapt (evolve) to the presence of drugs so that they do not die. They do this by changing their genetic make-up in a number of ways to stop the drug affecting them. Every time a bug grows, there is a chance that it or its offspring will adapt their genetic make-up, making them resistant to the drug. They are also able to “share” this ability to resist drugs, with their off-spring and other bugs even if they are of a different type.
Drug resistance is a natural process caused by bacteria, parasites, viruses or fungi developing to survive in their environment. In the fight against these bugs, some of them will eventually learn to adapt (evolve) to the presence of drugs so that they do not die. They do this by changing their genetic make-up in a number of ways to stop the drug affecting them. Every time a bug grows, there is a chance that it or its offspring will adapt their genetic make-up, making them resistant to the drug. They are also able to “share” this ability to resist drugs, with their off-spring and other bugs even if they are of a different type.
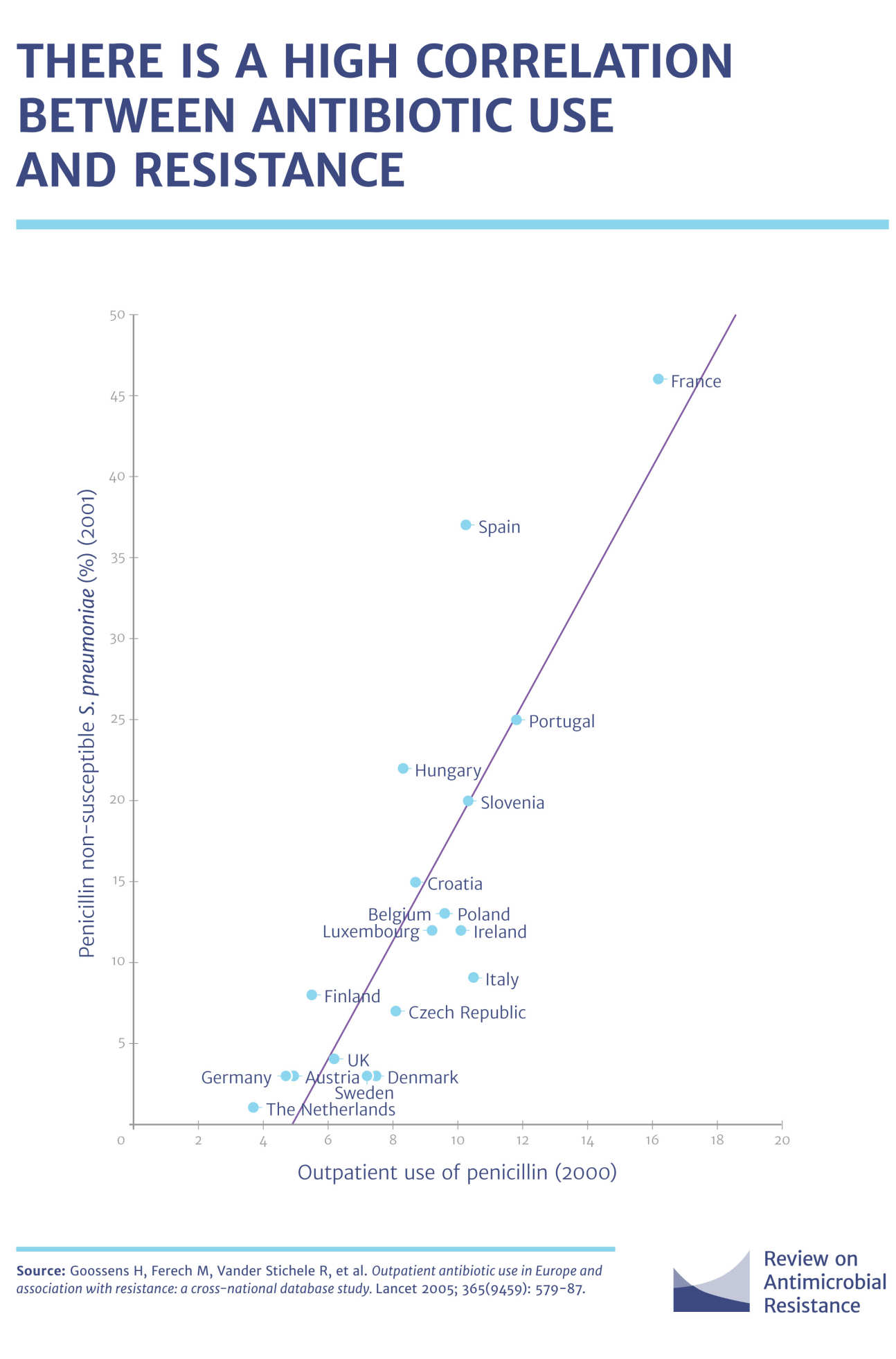
The development of drug resistance is, therefore, a risk or side-effect of using drugs in the first place. However, the development of resistance would not be such an issue if these drugs were only used sparingly as and when required and used carefully. However, since the discovery of penicillin in 1928 and its subsequent commercial production, antimicrobial drugs have NOT been used sensibly.
Drug resistance can develop from the use of drugs in humans, animals (pets and food-producing animals) or plants and for decades, antimicrobials have been used carelessly in human health, routinely in the production of livestock for food and in the environment.
Although work is needed to limit the use of antimicrobials in animals and the environment, the overuse and/or incorrect use of antimicrobial drugs in human health is thought to be the main contributor to resistance. As people do not complete their course of drugs, or miss doses, or take unnecessary antibiotics for coughs or colds against which they are not effective, the drug levels present are not enough to kill or prevent the growth of bacteria, thus allowing them to develop resistance and continue to multiply and grow. In the case of bugs, it is true to say that what does not kill them only makes them stronger.
How serious is the current situation with drug-resistant infections?
In July 2014, the UK Prime Minister commissioned a Review on Antimicrobial Resistance to analyse the global problem of rising drug resistance and propose concrete actions to tackle it internationally. The Review was jointly supported by the UK Government and Wellcome Trust, although was fully independence from both. The Review engaged widely with international stakeholders to understand and propose solutions to the problem of drug-resistant infections from an economic and social perspective, and produced its final report and recommendations in the summer of 2016.
The Report found that 700,000 people die of drug-resistant infections every year and that by 2050, an extra 10 million lives a year would be at risk due to the rise of drug-resistant infections - more than currently die of cancer if solutions are not found now to slow down the rise of drug resistance.
How do antibiotic resistance and drug-resistant infections affect me?
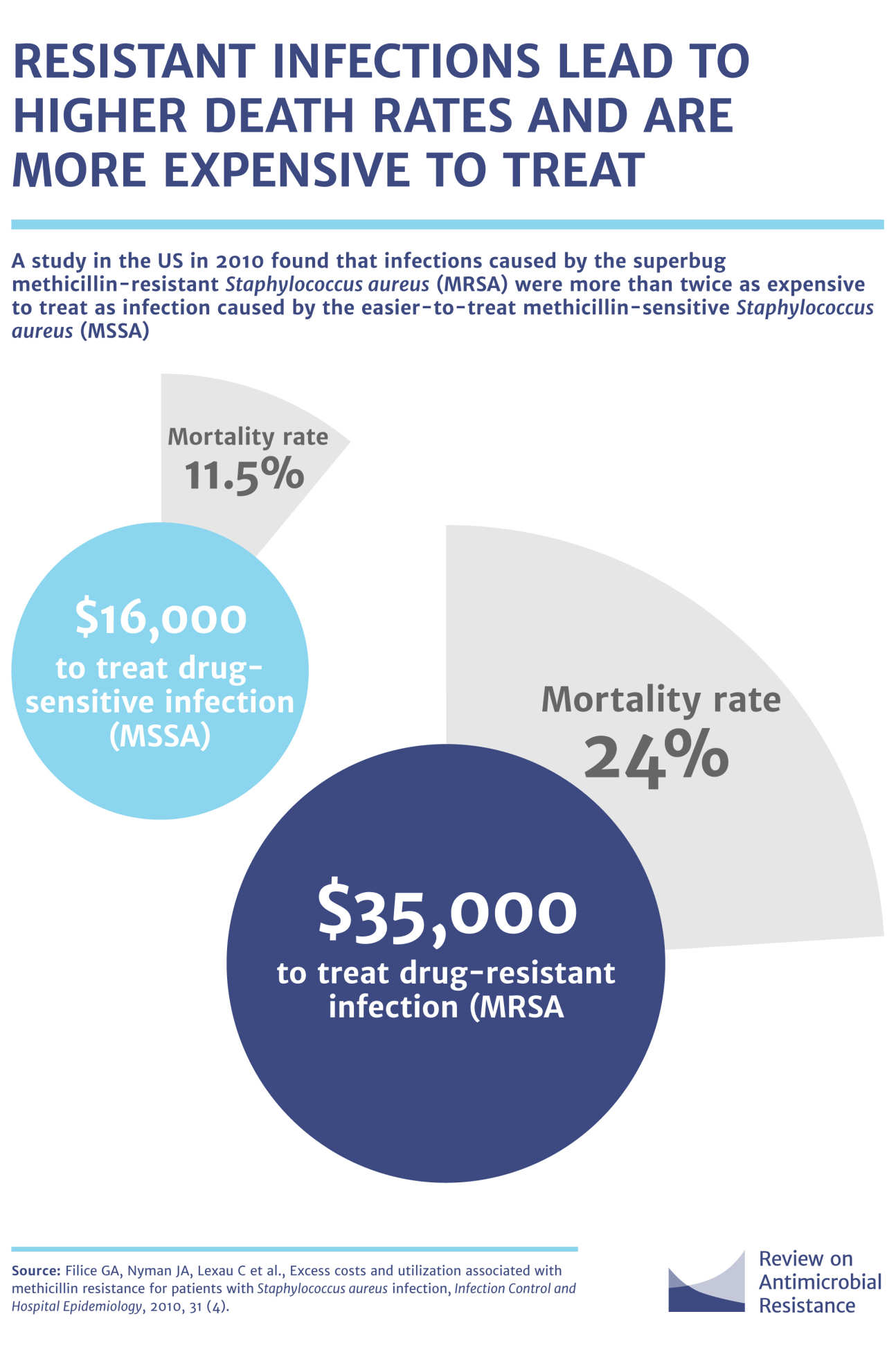
With drug resistance increasing, you may be more likely to catch a resistant bug. Infections caused by resistant bugs often fail to respond to the standard treatment, resulting in longer illness and a greater risk of death. For example, people with the drug resistant strain of Staphylococcus aureus, which is known as MRSA (methicillin-resistant staphylococcus aureus) are more likely to die than people with a non-resistant form of the infection which is a common source of severe infections in the community and in hospitals.
Resistance has also been found in bugs which cause common illnesses like urinary tract infections (Escherichia Coli), bloodstream infections/wound or surgical site infections and meningitis (Klebsiella pneumonia) and sexually transmitted diseases ( gonorrhoea and HIV). Increasing bugs are resistant to many types of drugs for example, extensively drug-resistant tuberculosis, a form of tuberculosis that is resistant to at least 4 of the core anti-TB drugs, has been identified in 105 countries.
Our ability to treat infections has allowed a revolution in modern medicine and the types of medical procedures which are now possible. Treatments for chest infections or urinary tract infections (UTIs), and surgery (from hip replacements to caesarian sections) are largely dependent on having drugs which can keep infection at bay. The government report “Ensuring access to working antimicrobials” [2014] concluded that: "antimicrobial resistance has the potential to send medicine back to the early 20th century, severely limiting the use of what are now considered basic and routine surgical procedures".
What is more, although investment is needed to stimulate the development of new classes antibiotics, this is not alone, a solution. Since bugs adapt and evolve, the time between the introduction of a new drug and the identification of resistance has become shorter and shorter. It is only by using our antibiotics more intelligently to preserve them for longer and by taking action to prevent the spread and transmission of existing drug resistance that we will improve the situation. Raising public awareness of the seriousness of this threat is vital together with an understanding of what we can all do to prevent drug resistance.
How do bugs become resistant to drugs?
There are several main ways in which bacteria, parasites, viruses and fungi can be resistant to drugs - known as 'mechanisms of resistance':
- by developing ways of making the drug useless. For example, some bacteria can produce a substance which stops penicillin working
- by developing ways of making it more difficult for the drug to attach itself to the bug making the drug less effective
- by becoming drug resistant by either making it more difficult for the drug to enter the person/animal/plant or by pumping it back out of them
Bugs such as bacteria can pass on resistance very quickly once the resistance has been developed. This means it is vital that we do as much as we can to prevent antimicrobial resistance.
Why is resistance such an issue now?
Antimicrobial drugs such as antibiotics are not only used in human health, they are also used in agriculture in the farming of animals and production of crops for food. Overusing these drugs in in each of these areas, together with the increased level of global travel and a failure to develop new antibiotics, are all contributing to the growth of drug resistant infections.
Human health
Doctors sometimes prescribe antibiotics to patients without an infection, or prescribe them to patients with viral infections for whom they do no good because diagnosis can be hard. , One study showed that doctors were much more likely to prescribe antibiotics for children if they felt that the parents expected them to - so parent pressure makes a difference. The US Centres for Disease Control (CDC) estimates that in hospitals half of the patients receiving antibiotics do not need them, resulting in overuse of antibiotics. Uncontrolled access to drugs over the counter or available to buy over the internet can also lead to them being overused or used for the wrong purpose.
Animal health
Drugs used to treat and prevent bacterial infections in animals belong to the same chemical groups as those used for humans. This means animals may acquire bacteria that are resistant to antibiotics also used to treat human infections. Since bacteria can share their resistant genes with other bacteria, this could result in a group of animals becoming resistant and people could then acquire resistant bacteria from direct contact with animals. Also, certain resistant bacteria that are associated with food consumption, such as Campylobacter or Salmonella, may be transferred from animals to humans through improperly prepared food.
Plant agriculture
Just as in human and veterinary medicine, drugs are used to control the spread of disease-causing bacteria in plants. Although the use of antibiotics in plant agriculture is strictly regulated, resistant strains of bacteria have started to develop. There are concerns that resistance developed in plant-based bacteria could spread to human-based bacteria, but more research is needed to fully understand the impact this might have. However, drug-resistant infections in plant-based bacteria make it more difficult to control the spread of blight (a symptom in reaction to infection by a pathogenic organism) and disease. In turn, this means that farmers produce less food for us to eat because more of their crop is lost to disease.
What can I do about drug resistant infections and antimicrobial resistance?
The seriousness of the threat caused my drug-resistant infections and the scale of its potential impact may make it seem like an impossible problem to solve. However, everyone can take simple measures that can help reduce its alarming spread. Please read more below, or go to our videos section to view brief films about what can be done, or download one of our printable factsheets
Practice proper hand hygiene
Proper hand hygiene is the best defence against the spread of drug-resistant bugs and the development of resistance to many drugs. You can help in the battle against drug resistance by ensuring you wash your hands properly:
- before, during and after making food
- before eating meals
- after using the toilet
- after coughing, sneezing or looking after someone who is ill
- after touching animals, raw meat or rubbish
Studies show that while many of us say that we already do practice proper hand hygiene, in reality, we often don't wash hands properly, or don't do it long enough (at least 20 seconds is recommended). Research has found that approximately one-third of men and two-thirds of women actually washed their hands after using the toilet, although 99% said that they had done so.
The US Centers for Disease Control recommend the use of alcohol hand gel, which is effective against many bugs. Using hand gel is also more convenient than washing with soap and water when you are out and about. Carrying hand gel with you in a pocket or handbag is a small but effective action you can take.
If you are a parent or guardian make sure that your children and their friends understand the importance of proper hand hygiene and know how to wash their hands properly. The e-bugs website is a great free resource for teaching children about hand hygiene, bugs and drug-resistant infections
In hospitals, GP surgeries and nursing homes hand hygiene is even more important due to the higher risk of person- to-person transmission of drug-resistant bugsIt is particularly important that staff and visitors wash their hands frequently with soap and water or use alcohol-based cleansers. If you do not see a member of staff washing their hands before treating you or whomever you are visiting, you should speak up and suggest that they wash their hands.
Reduce the use antimicrobial drugs, including antibiotics, when they are not needed
Even if a new drug developed, resistance would quickly emerge if we continued to use it. Therefore, changing the way we all use drugs is essential. It is estimated that more than half of antibiotics are unnecessarily prescribed for upper respiratory infections like cough and cold illness, most of which are caused by viruses. Also up to half of antibiotic use in hospitals is either unnecessary or inappropriate. Hospitals and other healthcare settings are working to address this, but you can help too by not requesting antibiotics and letting your doctor decide whether you need them or not. Your doctor will explain why they have not given you a drug and what else you can do to get better. Parents, in particular, have an important role to play; antibiotic prescription rates are highest in children and we know that doctors are more likely to prescribe antibiotics when pressured to by parents or guardians. Antibiotics cannot cure viral infections and will not help you get better from a cold. Alternative ways of treating infections, such as ensuring you have your vaccinations will also help reduce antibiotic use.
Ensure that drugs are always taken correctly
Improper use of antibiotics gives bugs the chance to develop resistance since the active ingredient of the drug is not high enough to kill them. To avoid this you should:
- only get medicines which are prescribed by trained people and registered places like your GP or Pharmacy rather than online where the drugs available may be sub-standard
- take your medicines according to the instructions without missing doses
- complete the full course of treatment, even if you feel better
- never use leftover prescriptions
- never share antibiotics with others
- manage your medicines properly – tools such as 'my medication passport' can be very helpful for everyone.
Talk about drug-resistant infections and teach others about them
Public awareness of the severity of drug-resistant infections needs to be increased. Everyone has a role to play by discussing drug-resistant infections with friends and family to ensure they are informed about the everyday things they can do to help prevent them. Anything which spreads the message is helpful, whether through Facebook, Twitter, Instagram or Snapchat or by becoming involved with a number of action groups, or awareness initiatives. You can also download any of our factsheets to use and share. If you have school age children please tell their teachers about the e-bug resources, which are designed to link to the curriculum and make them aware of the lesson plans we run and support.
Get involved in our research
You may also wish to become involved in our unit’s research, in which case please see our get involved section or contact us via email head.ops@imperial.ac.uk
What are we doing to tackle drug-resistant infections/antimicrobial resistance?
The problem of drug-resistant infections won’t be solved overnight. It will take focused global action to monitor and prevent the spread of drug-resistant infections and everyone, from governments to individuals, will need to play their part.
Global action plans
In April 2014 the WHO published “Antimicrobial resistance: global report on surveillance”. Following this report, there were a series of meetings between member states to discuss ways to develop and implement a global program for surveillance of drug-resistant infections in human health. There has also been international progress to tackle drug-resistant infections through government commitments to an AMR Global Action Plan 2015 which includes objective to improve the way antibiotics are used in food-producing animals.
UN Declaration 2016
On 21 September 2016, a landmark declaration was made where 193 countries agreed to combat antimicrobial resistance at the United Nations General Assembly.
The agreement followed a worldwide campaign led by the UK’s Chief Medical Officer Professor Dame Sally Davies and the UK’s Health Secretary Jeremy Hunt to highlight the threat posed to modern medicine by antimicrobial resistance.
Every signatory to the UN Declaration has agreed that drug-resistant infections must be tackled as a priority. The nations have committed to:
- develop surveillance and regulatory systems on the use and sales of antimicrobial medicines for humans and animals
- encourage innovative ways to develop new antibiotics, and improve rapid diagnostics
- raise awareness among health professionals and the public on how to prevent drug-resistant infections
The UN Secretary General will create a group including UN agencies to accelerate action and report back in two years. The UN Declaration furthers the promises made by G20 leaders in September 2016 to consider how to stimulate research and the development of new antimicrobial products.
Europe-wide action
The European Centre for Disease Prevention and Control (ECDC), founded in 2004, has prioritised drug resistance in its strategy. The ECDC coordinates the surveillance of drug-resistant infections, monitor the use of antimicrobial drugs and collect data on healthcare-associated infections (HCAI) across Europe. They also provide evidence-based guidance on the prevention and control of drug-resistant infections and HCAI, provide training on surveillance and control of drug-resistant infections and HCAI and support European countries in any activities related to drug-resistant infections, antimicrobial use and HCAI.
The ECDC coordinates the annual European Antibiotic Awareness Day on 18 November to raise awareness of the problem of drug resistance.
The UK strategy
In the UK, the Chief Medical Officer and Chief Scientific Officer to the Department of Health, Dame Sally Davies, made AMR the focus of her annual report in 2011. The report set out the scale of the threat of antimicrobial resistance and made the case for action. This was followed in 2013 by the Department of Health 'Five-year antimicrobial resistance strategy'. The strategy aims to:
- improve the knowledge and understanding of AMR
- conserve and steward the effectiveness of existing treatments
- stimulate the development of new antibiotics, diagnostics and novel therapies
As a result of this strategy, AMR has been added to the National Security Risk Assessment, the government’s long-term risk register. UK Health Security Agency (UK-HSA), the Department for Environment, Food and Rural Affairs (Defra) and the Department of Health are leading the implementation of the UK five-year antimicrobial resistance strategy. There have also been two government reports. The UK government Science and Technology Committee published its findings in the “Ensuring access to antimicrobials” report in 2013 and in 2016 an independent review on antimicrobial resistance called “Tackling Drug –Resistant Infections Globally: Final Report and Recommendations”, led by Lord O’Neill, published its final report. The report included ten recommendations for tackling drug-resistant infections across the globe. If you would like to read more, please see the Review on Antimicrobial Resistance.
These ten recommendations are:
- A massive global public awareness campaign
- Improve hygiene and prevent the spread of infection
- Reduce unnecessary use of antimicrobials in agriculture and their dissemination into the environment
- Improve global surveillance of drug resistance and antimicrobial consumption in humans and animals
- Promote new, rapid diagnostics to cut unnecessary use of antibiotics
- Promote development and use of vaccines and alternatives
- Improve the numbers, pay and recognition of people working in infectious disease
- Establish a Global Innovation Fund for early-stage and non-commercial research
- Better incentives to promote investment for new drugs and improving existing ones
- Build a global coalition for real action – via the G20 and the UN
The seriousness of the threat has resulted in an increase in research funding for drug-resistant infection research, including major joint investment from the UK Research Councils and through government agencies. Antimicrobial resistance was also voted by the public as the winning theme for the 2014 Longitude prize. The £10M prize will be awarded to whoever can solve the challenge to develop a diagnostic tool which will revolutionise the accuracy with which antimicrobials can be prescribed.
Antibiotic Research UK
ARUK is a UK charity dedicated to serving patients with affected by AMR and they are determined to offer as much support as we can to those affected. Find out more about how they are supporting people who have been either been diagnosed with an antibiotic-resistant infection themselves, or who have cared for someone who has.
Research at Imperial College
The Health Protection Research Unit in HCAI and AMR at Imperial College was established in 2014 to carry out research in this area and you can read about the work in each of our themes on our Research themes page. The College has also established the Antimicrobial Research Collaborative to bring together work from across the College which is relevant in tackling the threat of resistance.


