Our state-of-the-art multidisciplinary facilities help provide researchers and students the best collaborative and supported environment in which to learn and conduct novel and world leading research
All space within the department is shared, open and collaborative. It can fall into a number of subcategories, including office and laboratory types, research-specific space, core facility space and supportive space (PPE, Utility, etc.)
All of our research facilities and teaching spaces are staffed and supported by dedicated and highly skilled technical staff, providing an efficient and effective means of delivering key technologies and ensuring the maintenance of a skills repository, which is essential in delivering our leading international activities.
Facility charges and costing can be accessed here - Queries to the relevant facility technical staff member or the Technical Operations Manager.
PPEUtility

Personal Protective Equipment (PPE)
The department provides essential PPE consumables to all researchers within the department, including gloves, labcoats (and laundering), face masks etc. The PPE stores are based in level 6 Bessemer (B601a) and level 1 Bessemer. This is in addition to LEV (including soldering) extraction and Fume Cupboards for high hazard work.
Contact: Marta Garcia Bellmunt

Utilities
The Utility rooms are dedicated spaces for sterilizing (GM waste, Instruments and media), Ice machines, the 3D print water Jet station and glass washing for all researchers. More information here
The department also provides and supports 1 distilled/pure water system per floor for researchers to utilise.
Contact: Nana A. Asamoah-Danso
Departmental Core Facilities

Locations: B220, B114 Bessemer Building and RSM 4.22
Contact: Marta Garcia Bellmunt
INTERNAL LIMS
3D printing is a process of making three dimensional solid objects from a digital file by laying down successive layers of material until the object is created. The Bioengineering 3Dprinter facility offers 3 different types of 3Dprinters depending on the purposes and application of your project.

Objet 30 Pro
Based in Bessemer B220
A high end rapid prototyping machine that allows you to print with 8 different materials at the industry’s best print resolution.
- Tray dimensions 300 × 200 × 150 mm
- Cost: £15+ material used
- Offered as a service by Departmental technicians

Ultimaker 2
Based in RSM 4.22 and B220
An entry level 3D printer. It comes equipped with a heated build platform and can print at a resolution of up to 20 microns. It supports both PLA and ABS and can print at a speed of 300 mm/s.
- 20 microns layer resolution
- a wide range of materials (PLA, ABS, CPE, CPE+, PC, Nylon, TPU 95A)
- Tray dimensions 223 x 223 x 205 mm
- Free of charge, own materials supplied
Link to site ->

3D Bioplotter
Based in B114, Cell culture core facility.
The Bioplotter is a versatile rapid prototyping tool for processing a great variety of biomaterials for computer-aided tissue engineering (CATE), from 3D CAD models and patient CT data to the physical 3D scaffold with a designed and defined outer form and an open inner structure.
- Build volume 150 x 150 x 140 mm
- Cost £50 per booking 4 hour booking internal use (Departmental), including techncial assistance
- Cost £100 per 4 hour booking external use (non-departmental), including techncial assistance
Link to site ->
Location: B220, LG04 Lower Ground Floor Royal School of Mines
Contact: John Waldock, Departmental Mechanical Engineer
Russell Stracey, Workshop Supervisor
Shared with Earth Sciences and Materials departments the main function of the Royal School of Mines mechanical workshop is to provide mechanical engineering support for teaching and research. Working closely with research staff and students, technicians help to prepare samples and design, develop and manufacture prototype experimental equipment from a wide range of engineering materials for use in teaching and research. The activities performed are:
- CNC (Computer Numerical Control)
- machining Spark & Wire EDM (Electro Discharge Machining)
- Grinding of metals and ceramics
Bessemer B220 is a workshop which is primarily used for projects/timetabled work. The lab has been fitted out for tensil testing, 3D printing, laser cutting etc. but is also suited for small mechanical work (drilling, sawing, gluing etc). Students are able to reserve a bench for projects.

Location RSM 3.19, RSM 4.22/4.25 4th floor Royal School of Mines.
Contact: Paschal Egan, Tarik Malik
Niraj Kanabar (White City)
RSM 4.22 is an undergraduate electronic teaching laboratory which facilitates the practical aspects of modules as well as project work.
Research groups must utilise this dedicated electronics facility rather than solder in their own laboratory space (unless risk assessments and suitable fume extracts are purchased and appropriately maintained).

Dual Power Supplies

Reflow controllers

Reflow Station

Signal Generator

Soldering stations

Tensile Tester

Locations: B113 & B113a, B114 & B114a Bessemer Building.
Contact: Joel Eustaquio for Histolgy. Miguel Hermida for Tissue/Cell Culture, Microscopy, Bio and Laser safety and Cryopreservation. Marta Garcia Bellmunt for Genomics and Microbiology.

Tissue / Cell Culture
Containing all essential equipment for modern CLASS II primary and established cell culture use, including; Safety caninets, CO2 incubators, microscopes, centrifuges etc.

Histology
A dedicated space for Histology work open to all departmental researchers wanting to conduct in activities, such as; Staining, Embedding (cryostat and microtome sample preparation),Sectioning with cryostat and microtome and use of chemical fume hoods
•Cryostat NX70 and Bright Cryostats
•Microtome Leica RM2255
•Histocutter
•Embedding station and ovens

Genomics
A dedicated space for Molecular Genomics work open to all departmental researchers wanting to conduct in a molecular genomics activity, such as:
- RNA/DNA extractions
- Preparation of RNA/DNA
- PCR and Real time PCR
- Agarose gel electrophoresis
- Western blotting
- SDS PAGE
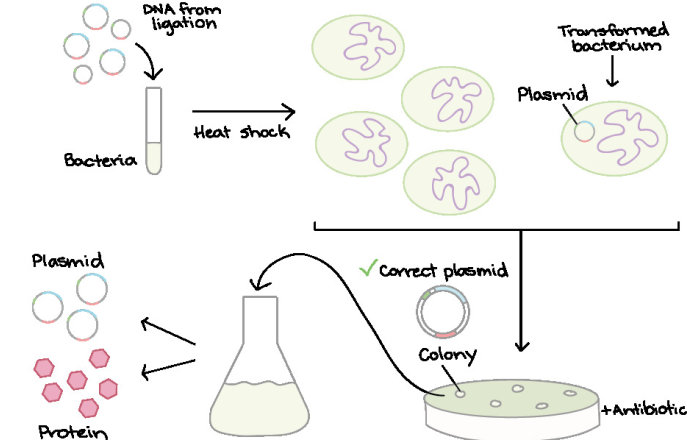
Microbiology
A communal space for work involving the transformation, cloning and culturing of bacterial cultures, for example for plasmid preparation and GM construction and testing
![]() Locations: B114a, B106 Bessemer level 1. TissueCyte: B306, Bessemer level 3
Locations: B114a, B106 Bessemer level 1. TissueCyte: B306, Bessemer level 3
Contact: Miguel Hermida
INTERNAL LIMS
The Bioengineering microscopy facility includes; Confocal, LCM, dissection, flourescence and light microscopy as well as highly specialised imaging and sectioning microscopes with the Histocutter (see Histology) and the TissueCyte 1000.
 TissueCyte 1000
TissueCyte 1000
An automated system from TissueVision Inc. capable of two-photon, sub-micron resolution imaging of entire small organs, ex vivo. A mouse brain, or similarly sized organ, can serially imaged over several days to generate three-dimensional data that can be used to map structures of interest such as cells, plaques or white/grey matter morphology.
PI charge out facility. Charged per hour of usage. Contact for more information.

Leica SP5
- 100 mW Argon laser 458, 476, 488, 496 and 514 nm
-
100 mW Argon laser 458, 476, 488, 496 and 514 nm
-
2 mW HeNe 594 nm
-
10 mW HeNe 633 nm
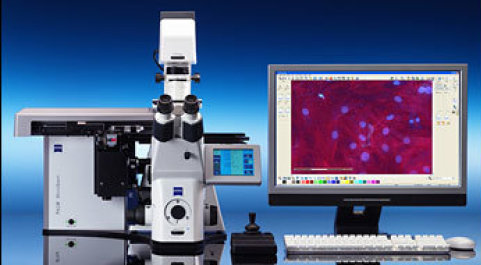
Zeiss PALM laser capture microdissection (LCM) microscope
Suitable for laser microdissection and analysis for DNA, RNA and protein isolation — whether from archive material or live cells. Applicable to cryosections, FFPE (formalin fixed and paraffin embedded) tissue
Software: PALM Robo v 4.6

Brunel SP105F with pE300 LED fluorescence microscope
Inverted fluorescence microscope;
- Transmitted illumination - 6v 30watt (bright field and phase contrast available)
- Objectives: 10x, 20x, 40x
- Fluorescence channels: UV1 - DAPI,
- B1 – FITC, G1 – TexasRED
- Fitted with Canon EOS 1200D camera
Core Facility categories
Engineering Facilities
Also known as tension testing, this is a fundamental materials science and engineering test in which a sample is subjected to a controlled tension until failure
Open for use under stupervision/training
Scientific stations
Availble on each bench station for use
Core utilities
The Bioplotter is a versatile rapid prototyping tool for processing a great variety of biomaterials for computer-aided tissue engineering (CATE), from 3D CAD models and patient CT data to the physical 3D scaffold with a designed and defined outer form and an open inner structure.
- Build volume 150 x 150 x 140 mm
- Cost £50 per booking 4 hour booking internal use (Departmental), including techncial assistance
- Cost £100 per 4 hour booking external use (non-departmental), including techncial assistance
Core facility equipment is:
- provided by academic shared cost and grant funding
- maintained and serviced utilising facility usage charges
- supported and managed by the departmental technical staff
