Meet 7 Imperial researchers working to end TB

World Tuberculosis (TB) Day held annually on the 24 March, marks the day in 1882 when the bacterium causing this disease, Mycobacterium tuberculosis, was discovered. Tuberculosis (TB) is one of the world’s leading causes of death due to infectious disease. This year’s World Health Organisation (WHO) theme is ‘Yes! We can end TB!’. The day aims to inspire hope and encourage high-level leadership, increased investments, faster uptake of new WHO recommendations, shorter all-oral treatment regimens for drug-resistant TB, accelerated action, and interdisciplinary collaboration to combat the TB epidemic. This year there is also a 2023 UN High-Level Meeting on TB.
At Imperial College London, researchers from across faculties, centres and units channel their expertise in genomics, public health, machine learning and engineering into tuberculosis research such as discovery research, diagnostics, treatment, policy advice, and national TB programmes across continents.
Meet seven of the Imperial researchers working to end TB.
1. India's national TB elimination programme
Professor Nim Arinaminpathy (School of Public Health) has always been interested in using mathematical modelling to solve problems within the public health sector and answering questions that have direct implications within it. His recognition of TB’s “very strong public health importance” led him to focus his research on the transmission and control of this disease in countries with a high burden. He works with governments and national TB control programmes to try and understand what their priorities should be. Currently, he’s working with India, which has the world’s largest TB burden, and Kenya, where HIV is a major driver of TB.
TB has a latent phase of infection, with some people subsequently developing active disease. “The infectious period can go on for months, if not years,” he says. Therefore, countries need to pull together very tight resources and figure out how to best control their TB epidemic.
This is where Professor Arinaminpathy’s work can help. With the use of modelling, he brings together the available evidence on how much TB burden a country has, who their vulnerable populations are, and how effective their interventions are. He can then help them prioritize which interventions would be most impactful in reducing TB incidence and mortality.
This year, Professor Arinaminpathy is working in India for World TB Day. Listen to the podcast to find out about the work he’s doing there, how antimicrobial resistance is affecting TB, and how he feels we can end TB.
TB control efforts were badly affected by COVID-19. With healthcare staff being diverted into the COVID-19 response and people not able to access care facilities, cases of TB were going undiagnosed. As a result, instead of the TB burden slowly declining over the last few years, it started to increase. The mathematical models produced by Professor Arinaminpathy and his team for the WHO were able to estimate how strongly these increases were occurring on a global level.
Interdisciplinary research is going to be absolutely critical in decreasing the TB burden.
The healthcare professionals and programme managers on the front lines must work with scientists to get the tools they need to manage this disease. As an example, Professor Arinaminpathy explained how TB is a curable disease, with most control programmes relying on a standardized six-month regimen - with which most cases of TB are curable - as well as focussing efforts towards getting this treatment to people on time. “But," he says, "we ultimately need a new vaccine against TB; one that can actually prevent people from getting infected or, even if they were infected, from developing the dangerous active form of the disease. To fulfil that whole chain, you need basic scientific research, you need clinical research, you need implementation research and mathematical modelling.”
In 10 years’ time, Professor Arinaminpathy hopes, “we would have an effective TB vaccine which would have been licensed for several years. I hope we will be working in earnest and that [there will be] new regimens for preventive treatment of TB and new tools that can help identify who would be most likely to develop TB. But also importantly, [I hope there will be] new ways in which the richer countries in the world can help mobilize resources in order to help low- and middle-income countries in their fight against TB. I hope as a community, we will be getting to grips with the ways in which we can combat this disease of poverty.”
2. Control the spread of drug-resistant TB
Like many bacterial infections treated with antibiotics, over time, mutations in the genome of Mycobacterium tuberculosis can give it the ability to survive treatment, therefore becoming drug resistant. Around half a million people suffered from drug-resistant tuberculosis in 2021, according to the WHO, and it continues to be a serious public health threat. The COVID-19 pandemic, which saw a rise inappropriate prescriptions of antibiotics, exacerbated this problem, with the estimated number of people with drug-resistant TB rising by 3.4% from 2020 to 2021. Combining genomics and machine learning is helping researcher Dr Leonid Chindelevitch, from the MRC Centre for Global Infectious Disease Analysis, understand the ways in which Mycobacterium tuberculosis mutates to develop drug resistance.
Dr Chindelevitch’s interest in medicine started when he was young, inspired by his grandmother, a doctor. Wanting to find a way to contribute to medicine in a non-clinical role, it was at graduate school that he discovered how he could combine his skills in computer science and mathematics with his interest in medical science. This was also where he identified tuberculosis as “one of the diseases that could really benefit from the use of computational tools such as machine learning, as well as genomics, for furthering our goals of controlling and perhaps ultimately eradicating it.”
Being able to diagnose any new tuberculosis infection quickly and accurately for susceptibility or resistance to available drugs allows the focus to be on interventions that do work. Treating tuberculosis disease adequately also means that the onward transmission from that patient to others significantly reduces, so it not only helps the patient but also those around them. Dr Chindelevitch’s research into the genomic determinants of Mycobacterium tuberculosis will help in diagnosis, to differentiate TB from viral fevers or HIV, which it often co-occurs with, especially in Sub-Saharan Africa. Genomics is also enabling researchers to determine what drugs the bacteria respond to and inform patient regimens so they can be as targeted as possible to the specific infection they have.
The future of tuberculosis research is really exploring how we personalise treatments and shorten regimens according to the genetic profile of the strain that’s impacting the patient.
Dr Chindelevitch collaborated on a paper, published last year, that introduced a WHO-endorsed catalogue of mutations to serve as a global standard for interpreting molecular information for drug resistance prediction.
Dr Chindelevitch understands that eventually eradicating tuberculosis may be difficult, but “what is achievable is significantly reducing the burden of disease that it causes.” He notes it is also a multifactorial problem, and efforts from researchers, clinicians, public health professionals, patients, and patient advocates are needed to reduce the stigma associated with the disease and raise awareness. He hopes we can “go back to focusing more on tuberculosis and put a stop to the overuse of antibiotics in situations where they are not appropriate. This can be achieved using diagnostics.”

3. Child and adolescent tuberculosis

Until recently, children have not been the focus of research in the field of tuberculosis, despite infections in children representing 10% of all tuberculosis cases worldwide.
Tuberculosis is the leading cause of death from infectious disease in all children globally so research into protecting children from infection and severe disease is vital. Imperial College London experts are looking at how children who are infected are managed in health care settings by exploring the use of new treatments and vaccines.
Dr Elizabeth Whittaker from the Department of Infectious Disease specialises in pediatric TB and how vaccination and co-infection in childhood can impact on the severity of the disease and its progression.
“My interest in tuberculosis started when I worked as a trainee doctor in London. I cared for a four-month-old infant who progressed rapidly from early TB to devastating TB meningitis, with subsequent life-changing severe brain damage within a couple of weeks. I was motivated to explore why young children are so susceptible to severe disease, and whether we could do anything to prevent this, such as with novel vaccines, or early diagnosis."
TB is a fascinating organism, with abilities to hide from the human immune system and avoid the normal immune responses to cause severe disease.
"My research now explores immune susceptibility to severe TB disease and other pathogens including Cytomegalovirus, or CMV. I look at whether co-infection with viruses may explain why some children experience more severe TB disease than older groups."
A key strategy to protect children from severe TB disease is through simple programmes of contact tracing and the use of 'chemoprophylaxis'. This is the use of cheap easily accessible antibiotics (isoniazid and rifampicin) to treat TB infection, to prevent progression to TB disease. Antimicrobial resistance against these two cheap, common, but effective antibiotics threatens the value of these programmes.
There is however hope for new ways to detect and diagnose TB in children as Dr Whittaker explains: “New host-based diagnostics (such as transcriptomic signatures) are on the brink of validation and development into point-of-care testing. Funding this next crucial step and making an affordable point-of-care test for low-resource settings could be a real game changer.
“I hope that in the future we could put even part of the resources that went into COVID research into TB research to advance rapid diagnostics, vaccines, and public health programmes, so that the research is thriving and effective in low-resource settings, where it is urgently needed.”
Dr James Seddon, Reader in Global Child Health at the Department of Infectious Disease works on a wide range of areas within child and adolescent tuberculosis.
His research explores how the disease develops in children but Dr Seddon also tries to improve detection of TB in children to help develop better treatments.
“My interest in TB came from clinical exposure at an early stage in my career. I spent a bit of time working for Médecins sans Frontières in Cote D’Ivoire and saw a lot of children with both TB and HIV. It became apparent that there was an urgent need for better diagnostic tests and better treatments.
“I am currently researching TB meningitis, which is a horrible type of TB that affects the brain and causes a great deal of mortality and morbidity. One area that both my colleague Dr Elizabeth Whittaker and I are interested in is the impact of puberty on the host immune response to TB. As adolescents progress through puberty, their response to disease changes, impacting risk and the type of TB they develop. We have a poor understating of this, and we are both working together to study this.”
Antimicrobial resistance is however making treatment of tuberculosis in children increasingly more difficult as Dr Seddon explains: “Although we are lucky to be in an age when new antibiotics are being developed, it is disheartening to see resistance develop just as quickly and the prospect of organisms that are resistant to all known antibiotics is a reality in some rare circumstances, making treatment impossible. However, the only way to completely prevent the development of antibiotic resistance is to not use antibiotics and so there is always a trade-off between responsible use and risk of resistance development.”
In the next 10 years, Dr Seddon sees a positive outlook for TB research, especially in areas with low resource. “I think that we will be in a far better place to integrate some of the most recent scientific advances into routine care, even in low-resource settings. A more personalised approach to care is I think possible, where treatments are tailored to the individual and their individual disease pattern.”
4. New national latent TB screening programmes

Professor Ajit Lalvani is Chair of Infectious Diseases, Founding Director of the Tuberculosis Research Centre and Director of the NIHR Health Protection Research Unit (HPRU) in Respiratory Infections, Imperial College London, and Honorary Consultant Physician at Imperial College Healthcare NHS Trust. His research aims to protect the health of the population through tackling the world’s most serious respiratory infections such as tuberculosis. He has over 25 years of experience conducting patient-based research, which has been translated into practical solutions such as the first rapid blood test for TB, new national latent TB screening programmes, and discovering the mechanism of action of BCG, the world’s most widely used vaccine. In the late 1980s while Professor Lalvani was a medical student, he worked with the homeless in central London, many of whom were suffering with TB.
Tuberculosis is a disease that disproportionately affects the underprivileged
This inspired Professor Lalvani to pursue research in it. He was also fascinated by how diverse outcomes from TB infection are determined largely by the body’s immune response. Around half of the people exposed to TB do not become infected so “discovering what immune responses in their lungs enable them to resist infection is a big challenge we are currently investigating and feels like the holy grail of TB research in humans, not least because the implications for developing new tools to tackle the global TB pandemic will be huge”.
Professor Lalvani was part of the team that created a new national latent TB screening programme with the UK Health Security Agency (UKHSA), which offers free latent TB testing and treatment to new entrants in primary care. It is the world’s first such programme and is designed to prevent imported latent TB infection carried by recent migrants from high TB burden countries. “Recently we have evaluated it and identified that it is highly effective, but its effectiveness could be substantially further improved if more new entrants participated in the programme and if the tests used for detecting latent TB were more highly predictive of who will actually progress from symptom-free latent TB infection to symptomatic active TB disease. We are working on both these aspects, the first being sociological and the second involving the application of cutting-edge immunology and genomics to develop a more predictive test (biomarker) for latent TB. We have made good progress on both fronts in the last few years.”
Having developed the world’s most advanced national programme for preventing imported latent TB, the team is now developing new approaches to stem the transmission of TB within the UK. To do this, they are developing new mathematical models to analyse the genome sequences of the TB bacteria from all TB patients in the UK. “UKHSA is the first national public health agency to sequence the genomes of TB bacteria from all TB patients and this collaboration is another example of the benefits of our multidisciplinary partnership with UKHSA through our HPRU,” he says.
“Implementing and evaluating the above national latent TB screening programme was only possible thanks to our seamless partnership with our national public health service (UKHSA). This is embodied in our NIHR Respiratory Infections Health Protection Research Unit which is a formal partnership, now almost 10 years old, between Imperial College London and UKHSA. Its remit is to tackle the major respiratory infections that threaten the health of the nation, particularly, TB, flu, RSV and emerging infections, such as COVID-19. To improve participation of new entrants in the screening programme we formed partnerships with community-based organisations with experience in working closely with recent immigrants, asylum seekers and marginalised groups.”
When asked about the achievement he is most proud of, Professor Lalvani reflected: “It’s hard to say what achievement I am most proud of after 30 years of active research, but it may be the discovery of how some people who have never previously been exposed to new lethal pandemic infections, including pandemic swine flu 2009 and COVID-19, are nonetheless protected from getting infected or becoming ill. In both cases, the answer lies in having pre-existing T-cells, induced by a previous infection, that cross-react to recognise and fight off the new pandemic infection, even though the person (and their T-cells) have not previously seen that new infection. The discovery provides the blueprint to develop universal vaccines against future pandemics.”

5. HIV-associated TB
Professor Robert Wilkinson (Department of Infectious Diseases and Founder of the Crick African Network) first became interested in TB in the early 1990s, when he was working at Northwick Park, a hospital in North West London, which was a referral centre for asylum seekers who had abnormal x-ray screens. “In the 1990s, there was a high prevalence and incidence of TB, particularly amongst Somali and Gujarati Asian immigrants, and I got very interested in them because no two cases were ever the same, and yet the treatment was, and still is to a large extent, completely standardized. I formed, and am still of, the belief that it needs to be more personalised.”
Only the minority of those exposed and infected with tuberculosis develop the disease, however, if a person has HIV, the likelihood of them developing TB is about 19 to 26 times greater. This led Professor Wilkinson to relocate with Wellcome support to South Africa where he has been involved in the research behind understanding the factors that are underlying the progression of, and tissue damage in, TB with a focus on HIV-associated tuberculosis. These infections remain acute healthcare problems in Africa, and he has been part of a joint programme with the University of Cape Town which is now in its 19th year.
He explains: “If an HIV-infected person developed TB, it became apparent that optimal management also required treatment for HIV to increase survival. But in certain circumstances that could lead to paradoxical deterioration of the TB, which is called immune reconstitution, inflammatory syndrome (TB-IRIS).” This occurs when there is an abnormal, excessive immune response against alive or dead Mycobacterium tuberculosis. An infected person develops lymph node swelling, worsening breathlessness or the onset of new meningitis, which represents TB infection of the brain: a condition that is particularly severe. “So, you have the ironic set of circumstances where two life-saving therapies may contribute to death.” One of the achievements of the programme has been to understand TB-IRIS better, in addition to preventing and treating it via the use of adjunctive steroid therapy. Via this work, Professor Wilkinson’s interests evolved to understand how to better treat TB meningitis and TB of the heart (pericarditis), another severe manifestation.
Through collaborations, the team also pioneered analysis of patterns of blood RNA molecules (transcriptomics) to determine if they’re specific to TB. A specific blood RNA signature was described that tended to resolve when patients were treated. There remains interest in whether patients not responding well can be spotted earlier. The team also combined transcriptomics with high-resolution functional and anatomical imaging. This helped them gain greater insight into early TB and how it advances. In turn, the imaging can be used to benchmark blood biomarkers of likely tuberculosis progression, thereby potentially advancing an important means to control TB: early detection and simplified treatment before it progresses, causes lung damage, and infects others.
More recently, the team have been involved in trials of a novel TB vaccine that shows encouraging signs it may arrest the progression of TB.
When asked about the impact of the COVID-19 pandemic on TB and HIV, Professor Wilkinson described the concern that, in addition to disruption of TB and HIV control, there could be biological interaction between the infections. In Western Cape Province, South Africa, there is a sophisticated electronic record linkage system that allowed healthcare encounters, whether they were at a clinic or GP or pathology lab or hospital, to be captured. From analyses of this valuable data, it became rapidly evident that HIV (especially untreated) and both treated and untreated TB were risk factors of death from COVID-19. This prompted Professor Wilkinson to instigate studies that simultaneously assayed the immune response to the pathogens. They found that immune antibody and T-cell response to COVID-19 was poor in people doubly- or triply- co-infected with co-existent TB or advanced HIV. However, mild COVID-19 infection did not seem to affect the immune response to TB which is encouraging.
“There are about 50,000 TB deaths a year in South Africa and it’s become again the commonest cause of death by infection.”
So, the funding, collaboration and energy that went into COVID-19 now needs to be directed towards TB.
South Africa also has a very significant problem with multidrug-resistant tuberculosis, defined as bacteria resistant to the first two frontline drugs, rifampicin and isoniazid. However, around 2006 it became more apparent that a more evolved form of drug-resistant TB, additionally resistant to fluoroquinolones and aminoglycosides, which were used as second-line drugs, was increasing in numbers. “The survival of HIV infected people with such extensively drug-resistant TB could be as little as a matter of weeks and months,” says Professor Wilkinson. Recent advances have shown that novel combinations of drugs such as bedaquiline, linezolid and delamanid greatly improve their outlook. “This together with widespread availability of antiretroviral therapy represents major treatment successes that have greatly improved the outlook for people with HIV-associated TB.”
In the next 10 years, Prof. Wilkinson would like to see drug regimens that are shorter and more easy to take, advances in early detection of progressive tuberculosis and its early treatment, and prevention by vaccination. He is actively working on each of these themes.
Celebrating World TB Day in a Cape Town Clinic
Celebrating World TB Day in a Cape Town Clinic
6. Futuristic rapid diagnosis

Ivana Pennisi is a PhD student who works in an interdisciplinary team that focuses on molecular diagnostic methods capable of detecting infectious disease-causing pathogens in a rapid and reliable manner.
Throughout her scientific career, Pennisi hoped her research would contribute to improving healthcare, particularly in countries where it’s difficult for patients to receive a fast diagnosis such as those in Africa. “I was given this opportunity when I joined my PhD project within the paediatric infectious disease group led by Professor Levin and Dr Kaforou, where they have been conducting an incredible number of studies on TB in children. It was impossible not to be inspired by their work, particularly when I was proposed to join Dr Rodriguez-Manzano and Professor Georgiou’s interdisciplinary research group at the department of Electrical and Electronic Engineering: a revolutionary team that has developed a microchip-based technology which could enable portable and prompt diagnosis at the point-of-need.” Speaking of this interdisciplinary approach, Pennisi noted: “Through the integration of infectious disease, molecular biology, mechanical engineering, and electrical engineering, we are working towards developing a test that will be capable of detecting TB from a drop of blood at the point of need.”
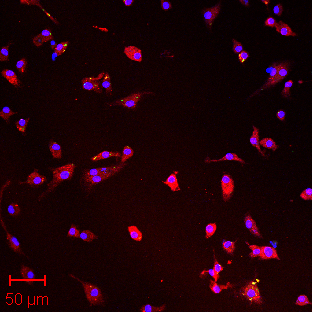
Lab-on-a-chip technology used in the EEE group to detect bacterial and viral targets.
Lab-on-a-chip technology used in the EEE group to detect bacterial and viral targets.
Together, using microchip-based molecular detection technology, the team aims to develop a rapid blood-based, diagnostic device test for point-of-care diagnosis based on host-gene signatures. This will offer early and accurate diagnosis of infectious diseases and be an important parameter in controlling the spread of TB.
“The diagnosis of infectious diseases such as TB in children is challenging," Pennisi. "Diagnostic methods for tuberculosis rely on the cultivation of the causative bacteria, which takes several weeks and usually requires invasive methods for the acquisition of appropriate samples, therefore there is a pressing need for a more accessible and accurate diagnosis of tuberculosis." The urgency for rapid diagnostics tools is further accelerated by the growing threat of drug-resistant tuberculosis. "Although there is an unprecedented demand for new antimicrobial molecules and regimens, rapid diagnostic tools are also urgently needed to reduce the time-to-diagnosis, contain outbreaks, and speed up the treatment process. Drug-resistant tuberculosis poses an obstacle that is surely challenging the tuberculosis research clock, intensifying the need for timely and verified data in order to make better-informed decisions to tackle the drug-resistant TB crisis."
Through ProtonDx, an Imperial start-up that was set up to deliver their portable molecular testing technology, the team intends to develop TB tests for both their existing and next-generation product platforms. Pennisi says: “The molecular diagnostics test, Dragonfly, which has recently received both UK and EU approvals for a five-virus respiratory panel, is our near-term solution for detecting tuberculosis. This would result in a portable and accurate TB test that can be used anywhere, including remote areas. As part of this research, we are also developing and testing our fully-automated, lab-on-a-chip platform known as Lacewing that builds upon the sample preparation and molecular assays created for Dragonfly, and will support early and accurate detection of tuberculosis."
Pennisi concludes: “In keeping with the positive and encouraging theme of World TB Day, I would like to extend my gratitude and full support to all researchers who have worked tirelessly over the past decades. It is important to realize that what we are doing is crucial. Even if the clock is ticking and there is massive pressure, if we continue to work together and share the important interdisciplinary tools available, we will undoubtedly achieve the important milestones required to end TB!"
Rising scientist day
Rising scientist day
Volunteering at a clinic in Gambia
Volunteering at a clinic in Gambia
Through the integration of infectious disease, molecular biology, mechanical engineering, and electrical engineering, we are working towards developing a test that will be capable of detecting TB from a drop of blood at the point of need.














