8 innovative ways Imperial is tackling malaria

World Malaria Day takes place on the 25 April this year, focusing on the theme of innovation. The Day not only provides an opportunity for the many different stakeholders involved in malaria research to highlight advances in prevention and control of the disease, but also the need for continued investment and political commitment towards these shared goals.
At Imperial College London interdisciplinary researchers from across all four of our faculties channel their expertise into malaria research via our Network of Excellence, as well as collaborating with researchers, policymakers and local communities across institutions and continents, including Target Malaria, the Digital Diagnostics for Africa Network, the Institute of Infection, and the MRC Centre for Global Infectious Disease Analysis.
Here are eight innovative ways Imperial is tackling malaria.
1. Mimicking natural immunity
The majority of malaria deaths are in children under five years old. However, the frequency of severe malaria and death decreases as children grow older because they gradually acquire immunity following repeated infections.
What if we could create vaccines that mimic this naturally acquired immunity? This is what Professor Faith Osier is investigating, in fine molecular detail. Creating vaccines for malaria is not simple, as the disease is caused by a parasite; a more complex organism than the virus that causes COVID-19 for example, and one that exists in multiple forms in different compartments of the human body.
So, while COVID vaccines target single proteins on the surface of the virus that are crucial for cell infection, the malaria parasite surface has many more proteins, making it is difficult to pin down which ones would stop it in its tracks. We can speed up the search for these proteins however by studying how natural immunity develops; by discovering which parts of the parasite the immune system is responding to in order to fight off the infection.
This involves studying what all the components of the immune system are doing and what they are reacting to – molecule by molecule. The hope is that with enough research, the ultimate vaccine could be created – one that mimics years of natural immunity in one shot.
Listen to Professor Faith Osier talk about why fighting malaria is so important, how her research is trying to create a vaccine that mimics natural immunity, and what the world needs to do to eradicate the disease.

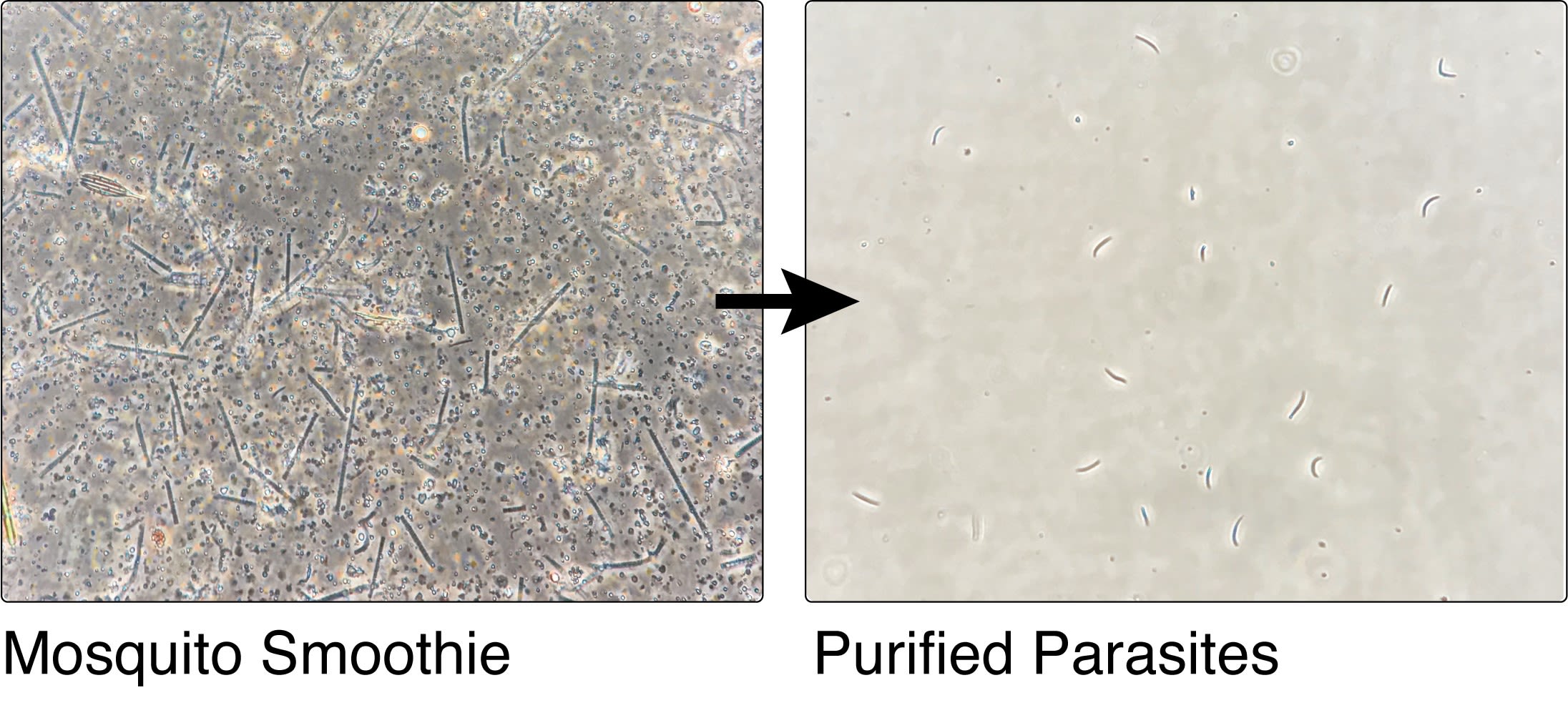
2. Making mosquito smoothies

There are existing malaria vaccines that provide moderate protection against the disease. However, they require several doses, with each dose requiring potentially tens of thousands of parasites at an early stage of their development, known as sporozoites. Extracting sporozoites for use in a live vaccine currently requires manual dissection of the mosquito salivary glands – miniscule structures behind the mosquito head – by a skilled technician.
Researchers from the Department of Life Sciences have developed a new, faster method for collecting pure malaria parasites from infected mosquitoes, which could increase both the scalability and efficacy of malaria vaccines. This new technique enables more parasites to be isolated rapidly with fewer contaminants and at far greater speed.
The method relies on making a ‘mosquito smoothie’ and filtering it, leaving a pure sporozoite product suitable for vaccination. The new method means no dissection is required.
Lead researcher Professor Jake Baum, from the Department of Life Sciences said: “Creating whole-parasites vaccines in large enough volumes and in a timely and cost-effective way has been a major roadblock for advancing malaria vaccinology, unless you can employ an army of skilled mosquito dissectors. Our new method presents a way to radically cheapen, speed up and improve vaccine production.”
Spinning a mosquito smoothie
3. Genetically modifying mosquitoes

One way to prevent the spread of malaria is to suppress populations of mosquitoes. Target Malaria is a not-for-profit consortium bringing together stakeholders across Africa, North America, and Europe with the shared goal of reducing malaria transmission by developing genetic technologies.
Recently, a team of researchers from our Department of Life Sciences, Polo GGB and Liverpool School of Tropical Medicine showed that ‘gene drive’ technology is able to suppress populations of malaria-carrying mosquitos in a year-long experiment mimicking natural environments.
Gene drive is a genetic phenomenon that occurs in nature and causes a selected trait to spread rapidly through a species via sexual reproduction over several generations. Gene drive can be harnessed by increasing the likelihood that a modified gene will be inherited by its offspring. Target Malaria is using gene drive to spread a modification affecting the mosquitoes’ reproduction (more male offspring or female infertility) to reduce the local populations of mosquitoes in order to stop malaria transmission.
Before any such mosquitoes are released, however, a lot of testing needs to be done. In the latest study, the team of researchers selectively targeted the specific mosquito species Anopheles gambiae and tested their modified mosquitoes in large indoor cages that simulated the natural environment. The study contained hundreds of mosquitoes of different ages, representing a natural and complex population structure, and was designed to mimic conditions in a way that cannot be achieved in small-scale testing facilities.
The large-cage trials are a major step on the journey to releasing gene-drive-carrying mosquitoes into the wild. The data obtained from the study will be invaluable to future modelling that aims to predict the spread of a gene drive in specific regions.
4. Engaging with local communities

Target Malaria brings together over 200 team members across eleven institutions in eight countries, with expertise ranging from entomology to anthropology, and molecular biology to modelling.
This requires innovation in stakeholder engagement, which is a key pillar of the project’s approach to responsible research, ensuring that the potential beneficiaries of the technology and those impacted by the work are at the heart of decision-making. Stakeholders range from local communities where the research takes place, to national authorities in the countries where teams work, and regional and international organisations with aligned interests, so engagement activities range from village meetings to international symposia.
Communities take part in scientific activities, such as mosquito collections, and in monitoring progress. When a new phase of work begins, local residents are asked whether they agree to planned activities, and agreement is repeatedly sought over time.
The focus is on the voice of those most affected by malaria, with a view that engaging stakeholders improves working processes, and the technology that’s ultimately developed.
One example of this work is in establishing a meaningful dialogue with stakeholders, which sometimes means overcoming linguistic challenges, such as those posed by appropriately translating scientific terminology employed by Target Malaria researchers in the field.
With this is mind, Target Malaria co-developed a series of local-language glossaries with communities living near field sites in Mali, Burkina Faso and Uganda. Teams set up a linguistic exercise to define key terms that didn’t have agreed-on translations in local languages, such as ‘gene drives’, ‘gene editing’ and ‘entomology’, finding appropriate translations that would empower communities to take part in dialogue on innovative vector control.
An initial glossary was tested on focus groups with community members, who were able to give their opinions, criticisms and feedback, making the proposed translations more appropriate to local context and cultural understanding. Team members across the project – including volunteers, field entomologists and those working in insectaries – were then all trained on these terms to ensure consistency.
As well as refining consistency in communication, the glossaries improve stakeholder understanding and ensure that consent and agreement – by individuals and communities – is effectively informed.
*
Target Malaria’s Approach to Stakeholder Engagement, an animation video, was produced in collaboration with Ryan Carter Images and Kikoto Animation studio.
5. Tracking mosquito behaviour

The parasites that cause malaria rely on mosquitoes, spending time in their guts and being passed on to humans via their bites. Understanding the behaviour of mosquitoes can therefore be crucial for predicting disease outbreaks.
For example, recent research led by Dr Marie Russell in the Department of Life Sciences showed how mosquitoes’ reactions to the predators that want to eat them can affect how malaria spreads in a community. With this knowledge, researchers say we can improve predictions of when and where there might be high numbers of human infections.
Mosquito behaviours are also important for another reason: predicting what might happen if we modify them, such as with gene drive technology (see number 3). A lot about the behaviour of these modified mosquitoes needs to be known before any are released. For example, Imperial researchers were recently involved in a project that tested whether modified (but not gene drive) mosquitoes can participate in normal behaviours alongside wild mosquitoes.
It’s also important to know how suppressing local populations of mosquitoes could affect the local ecosystem, with another study showing that no predator relies solely on the species of mosquito targeted by the modification, which is responsible for the majority of malaria cases in sub-Saharan Africa.

6. Providing cheap, portable diagnosis

One of the key ways to prevent disease spread is diagnosis. This poses a challenge in many areas as diagnosis usually requires analysing samples in a lab.
A team at Imperial developed a device to miniaturise the lab – to provide the functionality on a chip, which could provide portable and cheap diagnosis. The team is led by Dr Aubrey Cunnington from the Department of Infectious Disease and Professor Pantelis Georgiou from the Department of Electrical and Electronic Engineering, who are co-leads for the Digital Diagnostics for Africa Network, along with Professor Jake Baum from the Department of Life Sciences and Dr Jesus Rodriguez-Manzano from the Department of Infectious Disease.
The device required several innovations, including making sure the lab-on-chip technology can detect all the species of parasite that cause malaria in humans, and designing thousands of miniature sensors to detect the results of chemical reactions. The result is Lacewing: a device that can be linked to a smartphone app to provide the user directly with the results.
The team will next be taking their device out to African countries to check if it can work in a real-world setting, and if it can detect very low levels of parasites in people without malaria symptoms. Crucially, treating people like this early will help slow the spread of disease.
7. Modelling the best interventions

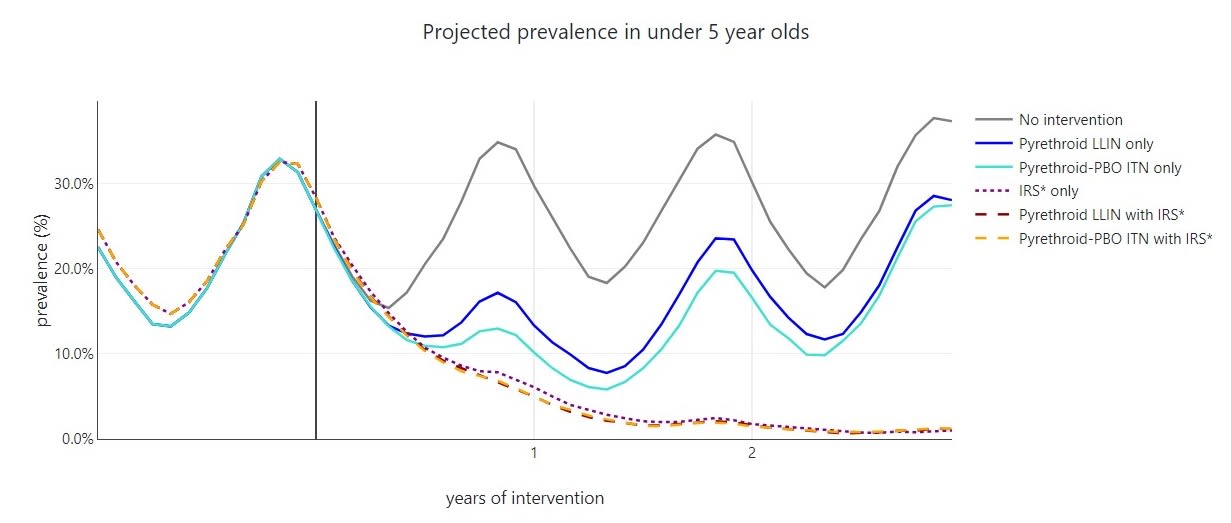
The outcome of MINT modelling given specific inputs including population size, seasonality, current malaria prevalence, how often mosquitoes like to bite, and levels of insecticide resistance. The graph shows the potential effect on the prevalence of infection in children under five years of age given a range of interventions including different insecticides
The outcome of MINT modelling given specific inputs including population size, seasonality, current malaria prevalence, how often mosquitoes like to bite, and levels of insecticide resistance. The graph shows the potential effect on the prevalence of infection in children under five years of age given a range of interventions including different insecticides
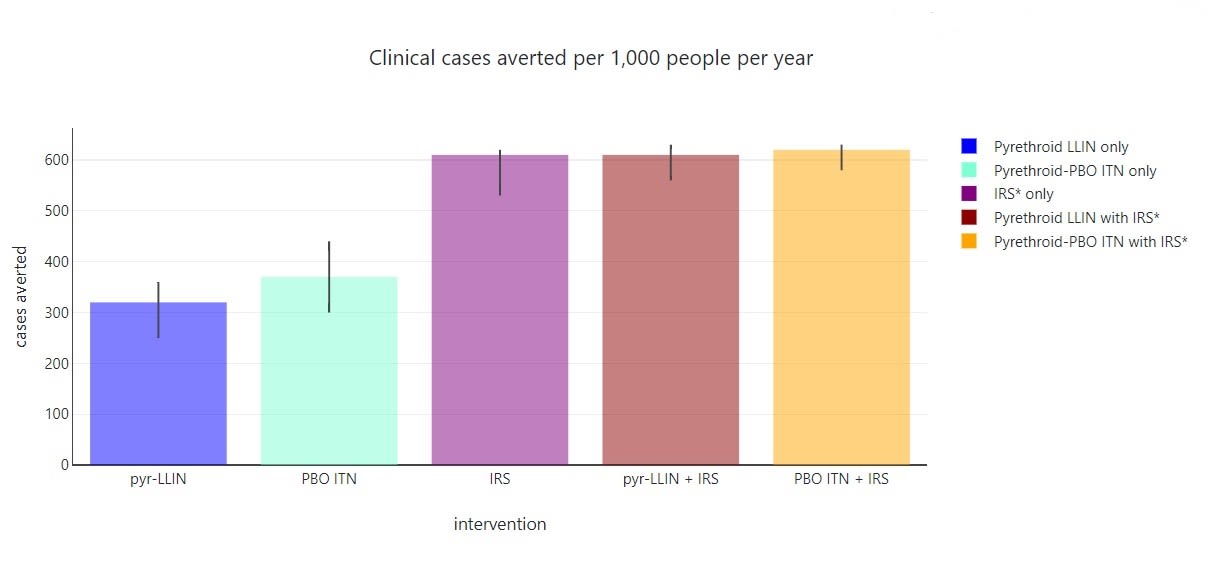
The outcome of the same MINT modelling inputs as above on clinical cases of malaria
The outcome of the same MINT modelling inputs as above on clinical cases of malaria
There are several methods of controlling local malaria cases, such as bed nets and insecticide spraying, but which ones are the best? There are many local factors to consider, including the environment, disease spread, and characteristics of the mosquitoes, such as whether they are resistant to certain insecticides.
A couple of trials of different combinations of nets and insecticides have been conducted, but the varying results suggest the local situation is very important. To overcome this limitation, researchers led by Dr Ellie Sherrard-Smith and Professor Tom Churcher from the MRC Centre for Global Infectious Disease Analysis at Imperial have created a model for predicting the best intervention for local situations.
Using the real-world data from the on-the-ground studies, they created an open-access online tool (Malaria INtervention Tool [MINT]) that aims to help policy makers identify the most cost-effective interventions for their setting across sub-Saharan Africa.
This is just one example of the kind of work the team does to better understand the transmission dynamics of malaria, using the resulting models to show how interventions can be optimally combined to reduce transmission, and ultimately to lead to local elimination of malaria.
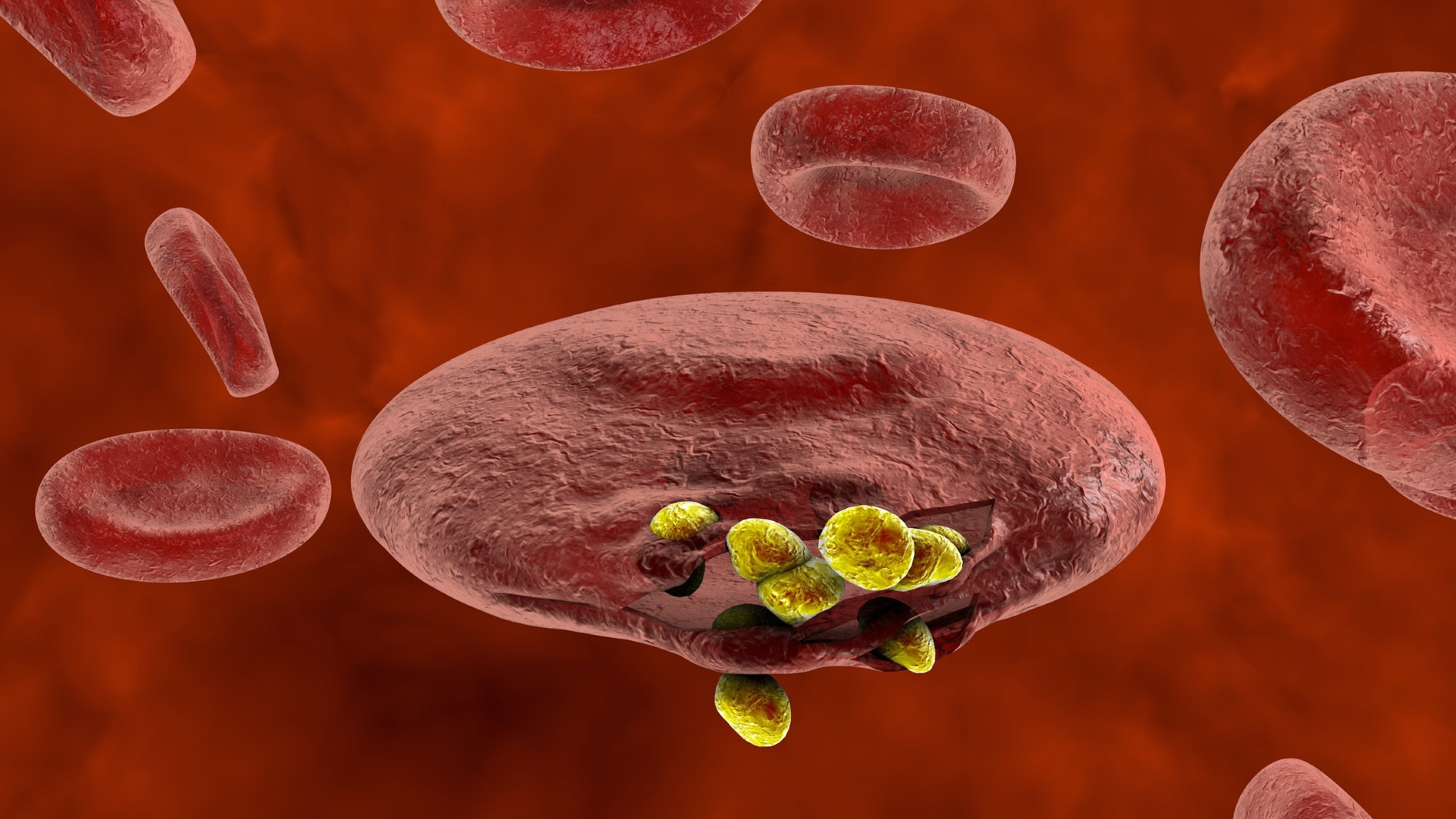
8. Monitoring parasite evolution
Fighting the parasite that causes malaria can be an arms race, with the parasite gaining resistance to drug treatments, for example. In order to stay ahead, we need to be aware of how the parasite is evolving.
Sickle haemoglobin – where human red blood cells adopt a different shape – has been associated with protection against malaria, since the parasite cannot as easily infect these cells. However, a team at Imperial recently discovered malaria genetic variants associated with the human sickle haemoglobin gene, suggesting these parasites may have adapted to overcome the protection offered.
Researchers sequenced the parasite genomes from over 3,000 infections in children, all of whom had severe symptoms of malaria, and compared their host and parasite genomes. They found that infections of individuals with sickle haemoglobin tended to be caused by a certain type of parasite. The researchers suggest that sickle haemoglobin in humans may have acted as a selective pressure on the parasite, driving it to adapt and leading to a population that can now infect people with sickle haemoglobin as well as those with normal haemoglobin.
From this study, further research into the biological function of these parasite variants and the mechanisms by which they interact with sickle haemoglobin can be investigated. Understanding these key questions could lead to new ways to protect against and treat malaria.
“This study highlights the importance of continually investigating this parasite so that we can be informed about how it adapts against selective pressures.”
Galvanising action
All these innovations will help us beat malaria. But perhaps the biggest innovation needed is the political will – getting our leaders to understand that we need urgent action to tackle malaria.
Professor Faith Osier has seen first-hand the devastating impact of malaria, which, for many of us, is a far-away problem. “There are images that will never leave me. I’ve seen the impact malaria has on a family when a child has died or become permanently disabled; the impact when the clinic is jammed with people lining up the whole day to get a simple malaria test. I feel the urgency with dealing with these problems because I have experienced them up close. I also appreciate the good health that I have been fortunate to have, and what that has enabled me to become. How many children and families does malaria rob of a future?
“Getting that political will and global determination to address a problem is so important. These problems are solvable and if people commit the resources that are needed, things will go a lot faster.”
“The closest comparison I can give is with COVID. When it hit Western countries we all felt it – the pain of lockdown, of losing someone, of being ill ourselves. We felt that urgency – that we needed a vaccine and we needed it yesterday, so we said let's do it, let's do everything that we can. For diseases that are far off, that sense of urgency is lost.
“Getting that political will and global determination to address a problem is so important. These problems are solvable and if people commit the resources that are needed, things will go a lot faster.”
Organisations are calling on countries to meet the Global Fund’s Seventh Replenishment target of at least USD$18 billion to fund malaria, AIDS and tuberculosis programs from 2024-2026. A fully replenished Global Fund will help accelerate progress and regain the ground lost during the pandemic.
Want to make a difference? You can support organisations like Malaria No More and their Zero Malaria campaign, or the RBM Partnership to End Malaria.
Keep up with all imperial's latest malaria-fighting innovations by following the Malaria Network on Twitter.