Smart diagnostics: the stepping stone in the pathway to malaria elimination
by Christo Hall

The next generation of smart malaria diagnostics hold the key to treating malaria effectively and assisting surveillance to stop its spread.
"Digital diagnostics have the potential to revolutionise the treatment and detection of malaria Aubrey Cunnington Head of Section and Reader in Paediatric Infectious Disease
Advances in technology have made malaria both preventable and treatable, yet over 400,000 people died from the disease in 2019, two-thirds of them children under the age of five and more than 90 percent of them in Africa. Ahead of this year’s World Malaria Day, Malaria Consortium and Imperial College Malaria Network take a look at the diagnostic innovations that could pave the way for malaria elimination.
Accurate, sensitive and affordable diagnostic methods are crucial to reducing malaria incidence and deaths. Conventional methods of malaria diagnosis used in low- and middle-income countries (LMICs), such as rapid diagnostic tests (RDTs) and lab-based methods such as microscopy, have been pivotal tools in achieving reductions in malaria cases. However, these methods have limitations that affect decisions about how best to treat a patient and how to deploy effective interventions to prevent further transmission.
Lab-based detection methods like microscopy are largely accurate, but are comparatively slow in providing results and require expensive equipment and training, which in LMICs means they are often only available in larger clinics and hospitals.
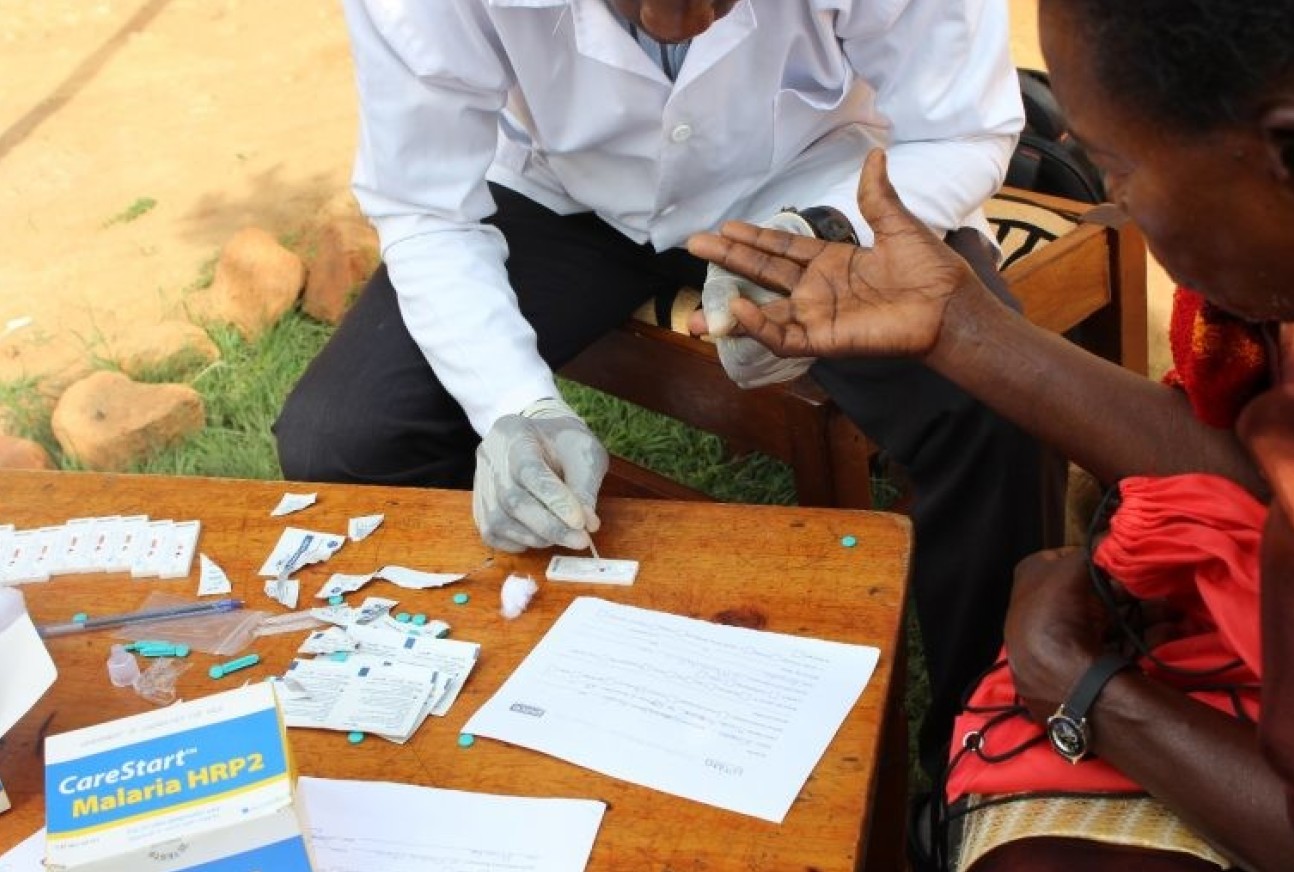 RDTs on the other hand are inexpensive and extremely fast at providing diagnosis (usually in under 20 minutes) and have therefore proved to be a useful tool for LMICs with a high burden of malaria. However, they are not sensitive enough for those with low-level infection without symptoms and there are increasing concerns over genetic mutations in the malaria parasite that mean the tests are unable to detect malaria in a patient. “In some areas, parasites have gained mutations in the genes which encode the protein detected by rapid diagnostic tests, meaning that the diagnostics no longer work in some patients. This is one of the first ever cases of a disease evolving resistance to a diagnostic, which emphasises the continual need for new digital tests that can detect new parasite targets to keep up with parasite evolution,” explains Dr Lucy Okell, Imperial College Senior Lecturer.
RDTs on the other hand are inexpensive and extremely fast at providing diagnosis (usually in under 20 minutes) and have therefore proved to be a useful tool for LMICs with a high burden of malaria. However, they are not sensitive enough for those with low-level infection without symptoms and there are increasing concerns over genetic mutations in the malaria parasite that mean the tests are unable to detect malaria in a patient. “In some areas, parasites have gained mutations in the genes which encode the protein detected by rapid diagnostic tests, meaning that the diagnostics no longer work in some patients. This is one of the first ever cases of a disease evolving resistance to a diagnostic, which emphasises the continual need for new digital tests that can detect new parasite targets to keep up with parasite evolution,” explains Dr Lucy Okell, Imperial College Senior Lecturer.
 One such digital test, Lacewing, a handheld electronic device developed by a team of scientists at Imperial College London, led by Dr Pantelis Georgiou, uses a small blood sample from a patient to provide diagnosis in around the same time as an RDT, sensitivity that exceeds microscopy and parallels the most advanced lab-based detection methods. According to Dr Aubrey Cunnington, Head of Section and Reader in Paediatric Infectious Disease at Imperial College London, digital diagnostics like Lacewing have “the potential to revolutionise the treatment and detection of malaria as they can detect all of the malaria parasite species and even low levels of parasite that cannot be seen under a microscope. Some devices have the ability to detect drug resistance mutations and other causes of illness, which gives a clinician an advanced picture of the infection.”
One such digital test, Lacewing, a handheld electronic device developed by a team of scientists at Imperial College London, led by Dr Pantelis Georgiou, uses a small blood sample from a patient to provide diagnosis in around the same time as an RDT, sensitivity that exceeds microscopy and parallels the most advanced lab-based detection methods. According to Dr Aubrey Cunnington, Head of Section and Reader in Paediatric Infectious Disease at Imperial College London, digital diagnostics like Lacewing have “the potential to revolutionise the treatment and detection of malaria as they can detect all of the malaria parasite species and even low levels of parasite that cannot be seen under a microscope. Some devices have the ability to detect drug resistance mutations and other causes of illness, which gives a clinician an advanced picture of the infection.”
In many LMICs, large proportions of the population do not have access to health facilities. Digital health platforms, like Malaria Consortium’s upSCALE in Mozambique, are helping to overcome this problem by providing health workers in local communities with a smartphone and tablet app that assists with patient registration, diagnosis and advice on treatment and referrals, as well as logging stock levels of drugs and equipment.
“Data entered through the app is captured in the District Health Information System at district, provincial and national level and the ability to analyse local disease-specific trends in near real-time improves resource allocation,” explains Dr Maria Rodrigues, Mozambique Country Director at Malaria Consortium. “Digital technology is already helping to overcome limited access to primary healthcare for rural populations and weak linkages between health workers and national health information systems but there are still huge gaps, especially when it comes to accessing the kinds of diagnostic technology available in hospitals.”
This gap could be filled by simple, portable and inexpensive digital devices like Lacewing, leading to a step change in how malaria and other infectious diseases are monitored and treated, effectively bringing the laboratory to the patient. “Digital diagnostics can improve health care in resource-limited settings by making available highly sensitive and reliable diagnostic tests at the most peripheral level and by registering clinical cases directly into the national surveillance system,” explains Professor Umberto D’Alessandro is Director of the Medical Research Council Unit The Gambia (MRCG) at London School of Hygiene and Tropical Medicine. “The latter would be able to generate real-time statistics, for example the number of cases of a specific disease, that could be used to rapidly distribute resources where these are most needed.”
Malaria Consortium’s experience and expertise in implementing malaria programmes in partnership with Ministries of Health means we are well placed to advise on how digital diagnostic tools like Lacewing can be integrated in, and add value to, National Malaria Elimination Programmes. Craig Bonnington, Head of Technical – East and Southern Africa at Malaria Consortium sees the potential these tools could have in practice: “The ability to detect asymptomatic infection, diagnostic and drug resistance combined with incorporation of this data into digital surveillance systems has the potential to change the trajectory of elimination projects across the continent with tailored context specific interventions.”
The huge potential of Lacewing and other digital diagnostics lies in their clinical and epidemiological application and simplicity. Alongside the portable device itself, the Lacewing platform contains a rechargeable battery, and the test can be performed through an app on a smartphone. “While the device will never get down to the price of an RDT, it can be mass produced cheaply and the technology and advantages it offers should be comparable to the results of expensive lab-based instruments,” Miss Kenny Malpartida-Cardenas, Research postgraduate in Pantelis Georgiou’s research team at Imperial College.
"Not only can we know if a patient has malaria but we can also capture a myriad of other diagnostic readings that might tell us of other co-morbidities, co-infections or susceptibility issues" Jake Baum Professor of Cell Biology and Infectious Diseases
As we know from our experiences with COVID-19, knowledge of transmission is vital to control infections. The more we know and the quicker we know it, the more effective localised interventions can be. National malaria elimination programmes are impeded by incomplete information about the number of infections, this barrier is antagonised by insufficiently sensitive diagnostics.
Improved and better integrated surveillance systems will ultimately not only improve outcomes for malaria but improve health outcomes more widely and build more resilient health systems better able to cope with events like the COVID-19 pandemic. “To make significant and meaningful progress in reducing the number of deaths from malaria, we need to move away from a one size fits all paradigm. The next generation of digital tools and diagnostics represents a significant breakthrough that will transform our malaria intervention and surveillance toolbox,” says Arantxa Roca-Feltrer, Malaria Consortium’s Head of Surveillance, Monitoring and Evaluation. “We need much better integration of community level data to inform and support surveillance efforts, which are still missing in many countries. Surveillance that brings fragmented information systems together and helps in understanding how malaria incidence is affected by, and impacts on, the bigger picture.”

The data that digital diagnostics are able to capture has the potential to inform and empower discrete interventions that considerably contribute to the elimination of malaria and aids timely decision-making at all levels including the health facility and community levels. Jake Baum: “The power of digital diagnostics is that they enable both mapping of infections in space and time and provide fine granularity of data to clinicians and healthcare workers. It means not only can we know if a patient has malaria but we can also capture a myriad of other diagnostic readings that might tell us of other co-morbidities, co-infections or susceptibility issues that need to be taken into account. Not only this, the data can also give us location, identification and, as we build up a centralised picture, region by region stratification that allows local and national authorities to roll out the right strategies for elimination in their region. In regions where populations are remote or not on any map, geolocation of point-of-care diagnosis becomes extremely powerful.”
This work demonstrates the potential the power of inter-disciplinary collaborations and partnerships and the potential for finding new ways to tackle the evolving challenges of diagnosing, treating and preventing malaria. All contributors to this article are part of the Digital Diagnostics for Africa Network, a UKRI-funded international community of international experts from 15 partner organisations spanning the public, private and third sector and all sharing the vision that “digital diagnostics can transform healthcare in Africa.” Continuing to invest in research and development, and scaling up innovations such as real-time data and new technologies are critical to making meaningful progress towards malaria burden reduction and elimination in multiple settings.
This article was produced in collaboration with Malaria Consortium.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Christo Hall
Department of Life Sciences