Gene therapy for inherited blindness produces promising first results
by Sam Wong
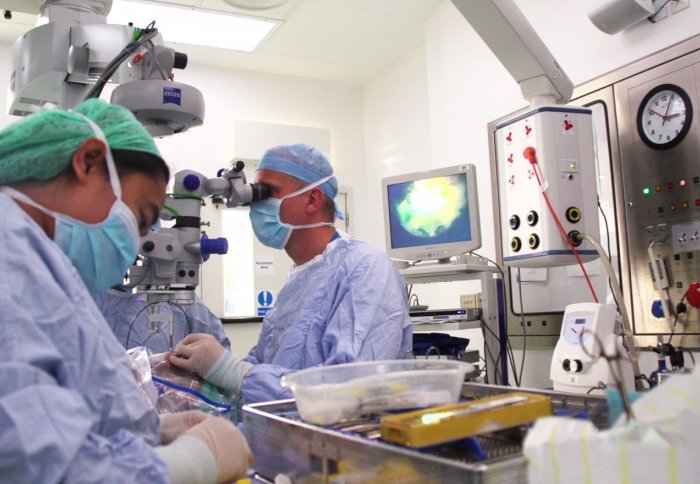
The first clinical trial of a gene therapy for an inherited cause of progressive blindness called choroideremia has shown promising initial results.
The treatment aimed to get the gene therapy into the cells in the retina of the eye without causing damage. After six months, however, the patients showed improvements in their vision in dim light and two of the first six patients enrolled were able to read more lines on the eye chart.
The results surpassed the expectations of the researchers leading the study from Oxford University and Imperial College London.
Professor Miguel Seabra, whose research at Imperial College London identified the protein involved in choroideremia, says: “My team has spent 20 years trying to understand choroideraemia and develop a cure, so to finally see the rewards reaching patients is extremely gratifying, both for us and the families who supported our research.”
A total of nine patients have now had one eye treated with the gene therapy in operations at the Oxford Eye Hospital, part of the Oxford University Hospitals NHS Trust. The therapy is given in one eye to allow comparison with progression of the disease in the other eye.
Results at six months are now reported for the first six patients in The Lancet medical journal. The first patient to have the operation has now been followed up for over two years. Based on the success of the treatment in the first six patients, three more have recently been tested at a higher dose.
Professor Robert MacLaren of the Nuffield Laboratory of Ophthalmology at the University of Oxford, and a consultant surgeon at the Oxford Eye Hospital and honorary consultant at Moorfields Eye Hospital, led the development of the retinal gene therapy and this first clinical trial.
“‘It is still too early to know if the gene therapy treatment will last indefinitely, but we can say that the vision improvements have been maintained for as long as we have been following up the patients, which is two years in one case,” he said.
The first patient to be treated, Jonathan Wyatt, 65, said: “My left eye, which had always been the weaker one, was that which was treated as part of this trial... Now when I watch a football match on the TV, if I look at the screen with my left eye alone, it is as if someone has switched on the floodlights. The green of the pitch is brighter, and the numbers on the shirts are much clearer.”
Wayne Thompson, 43, an IT project manager in Staffordshire, was treated in April with a higher gene therapy dose as part of the subsequent trial: “One night in the summer, my wife called me outside as it was a particularly starry evening. As I looked up, I was amazed that I was able to see a few stars. I hadn’t seen stars for a long, long time... For a long time I lived with the certainty of losing vision. Now I have uncertainty of whether the trial will work, but it is worth the risk.”
Choroideremia is a rare inherited cause of blindness that affects around one in 50,000 people. The first signs tend to be seen in boys in late childhood, with the disease slowly progressing until vision is lost. There is currently no cure. It is caused by defects in the CHM gene on the X chromosome, which explains why it is almost always boys that are affected. Without the protein produced by the CHM gene, pigment cells in the retina of the eye slowly stop working, then die off. As the disease progresses, the surviving retina gradually shrinks in size, reducing vision.
The gene therapy uses a small, safe virus to carry the missing CHM gene into the light-sensing cells in the retina. In an operation similar to cataract surgery, the patient’s retina is first detached and then the virus is injected underneath using a very fine needle.
The aim is for the CHM gene, once delivered into the cells of the retina, to start producing protein and stop the cells dying off. “If we were able to treat people early, get them in their teens or late childhood, we’d be getting the virus in before their vision is lost,” said Professor MacLaren. “If the treatment works, we would be able to prevent them from going blind.”
This is the first time that a gene therapy has targeted the principal light-sensing cells in the retina, known as photoreceptors. This approach has relevance for other, far more common causes of blindness where these light-sensing cells are affected, such as retinitis pigmentosa and age-related macular degeneration.
The phase I clinical trial is funded by the Health Innovation Challenge Fund, a partnership between the Wellcome Trust and the Department of Health.
The research has received additional support from the National Institute for Health Research Oxford Biomedical Research Centre and the charity Fight for Sight, the main UK charity that funds pioneering eye research to prevent sight loss and treat eye disease.
Patients have come from Moorfields Eye Hospital, the Manchester Royal Eye Hospital and the University of Southampton.
Reference: R.E. MacLaren et al. ‘Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial.’ The Lancet, Early Online Publication, 16 January 2014. doi:10.1016/S0140-6736(13)62117-0
Based on a news release from Oxford University
To enquire about taking part in future trials, please contact the Nuffield Laboratory of Ophthalmology at Oxford. enquiries@eye.ox.ac.uk
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Sam Wong
School of Professional Development