Parkinson's disease protein plays vital 'marshalling' role in healthy brains
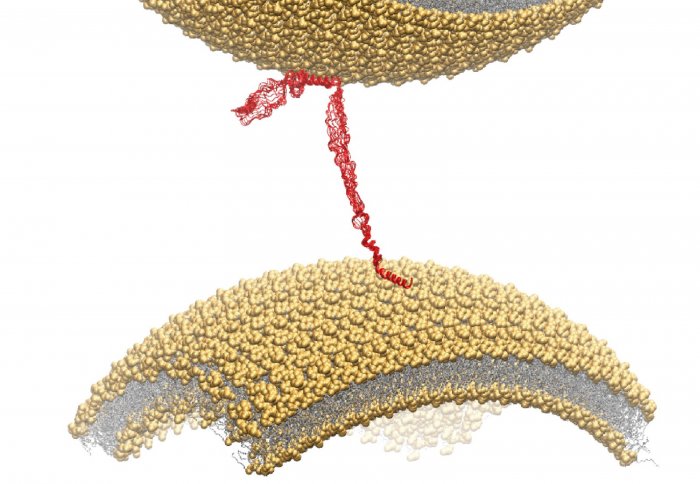
Alpha-synuclein (red) acts as a bond between cellular transporters in healthy brains
Researchers have uncovered the normal function of a protein associated with Parkinson's disease, giving clues about what happens when it malfunctions.
By showing how the protein - called alpha-synuclein - works in healthy patients, the study offers important clues about what may be happening when it malfunctions and people develop the disease.
While malfunctioning alpha-synuclein has long been recognised as a hallmark of Parkinson’s disease, its role in healthy brains was not properly understood until now. The new study, carried out by researchers at Imperial College London and the University of Cambridge shows that the protein plays a vital role in signalling in the brain.
If you remove part of a machine, you need to know what it is supposed to do before you can understand what the consequences of its removal are likely to be.
– Dr Alfonso De Simone
The research team found that alpha-synuclein regulates the interaction between synaptic vesicles, which are small containers of neurotransmitters – the signalling chemicals of the brain. Alpha-synuclein helps to marshal the correct number of vesicles into the right position to release their neurotransmitters across junctions between nerve cells, known as synapses.
Two different regions of the protein were found to have membrane-binding properties that mean it can attach itself to vesicles and hold some of them in place, while others are released.
Significantly, the researchers also tested mutated forms of alpha-synuclein that are linked to Parkinson’s disease. These were found to interfere with the same mechanism, compromising the ability of the brain to signal between neurons. The research is published today in Nature Communications.
Machine learning
Study co-author Dr Alfonso De Simone from the Department of Life sciences at Imperial said: “If you remove part of a machine, you need to know what it is supposed to do before you can understand what the consequences of its removal are likely to be.”
“We have had a similar situation with Parkinson’s disease; we needed to know what alpha-synuclein actually does in order to identify the right strategies to target it as a therapeutic approach to Parkinson’s.”
Giuliana Fusco, a Chemistry PhD student from the University of Cambridge who has now joined the team at Imperial, carried out the main experiments underpinning the research. She said: “It was already clear that alpha-synuclein plays some sort of role in regulating the flow of synaptic vesicles at the synapse, but our study presents the mechanism, explaining exactly how it does it.”
“Because we have shown that mutated forms of alpha-synuclein, which are associated with early onset familial forms of Parkinson’s disease, affect this process, we also now know that this is a function that may be impaired in people who carry these mutations.”
Compromised role
The research suggests that in some familial cases of early onset Parkinson’s disease, because alpha-synuclein malfunctions as a result of genetic alterations, the protein’s marshalling role is compromised.
One of the trademarks of Parkinson’s disease, for example, is an excess of alpha-synuclein in the brain. In such circumstances, it is possible that too much binding will take place and the flow of vesicles will be limited, preventing effective neurotransmission.
The study involved lab-based experiments in which synthetic vesicles, modelling the synaptic vesicles found the brain, were exposed to alpha-synuclein.
Using nuclear magnetic resonance spectroscopy, the researchers examined how the protein organised itself structurally in relation to the vesicles. To verify the findings, additional tests were then carried out on samples taken from the brains of rats.
-
'Structural Basis of Synaptic Vesicle Assembly Promoted by α-Synuclein' is published in Nature Communications.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Hayley Dunning
Communications Division