The new "super filters" that could revolutionise carbon capture
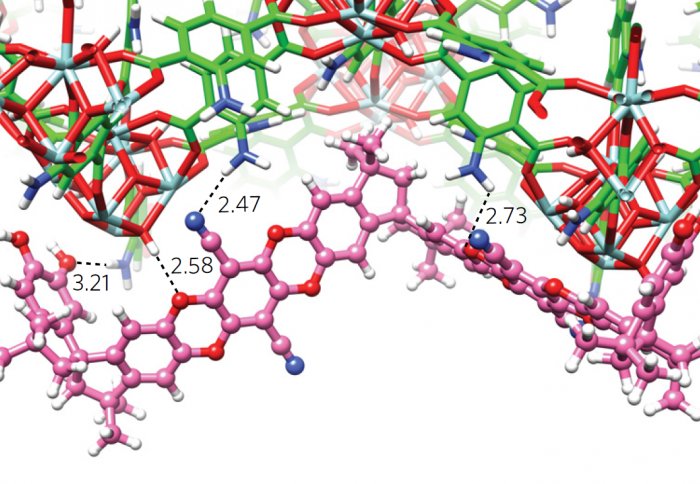
Molecular simulation of mixed matrix membranes. Credit: Nature Publishing Group
New research into the development of high-performance membranes for carbon capture applications has been published in Nature Energy.
Read the full article online here
The paper details research undertaken by Kyoto University's Institute for Integrated Cell-Material Sciences (iCeMS), the City University of Hong Kong and Imperial College London into the next-generation of separation membranes: highly-permeable and selective Mixed Matrix Membranes (MMMs) that could potentially revolutionise carbon capture and storage (CCS) technology.
Conventional CO2 capture technologies, such as amine absorption process, are complex and significantly energy-intensive. Membrane separation processes are used in industry in various contexts, from water purification to natural gas purification, with higher energy efficiency and lower environmental impacts.
However, a key problem faced by large-scale membrane-based carbon capture projects is the slow gas throughput of commercial membranes, which typically leads to increased operation costs.
"Until now polymer membrane technologies for gas separation applications have not been up to the task, because there is a poor trade-off between low gas permeability and high degree of selectivity for separation of molecules" explains Professor Easan Sivaniah from Kyoto University's Institute for Integrated Cell-Material Sciences (iCeMS), lead author of the study.
"Gas permeability is the intrinsic property indicating how fast gas molecules pass through a membrane, and the selectivity calculated as the ratio of permeability of two gas species determines the capability to separate gases. Novel membrane materials with both high permeability and high selectivity are the key for large scale application of membrane technology in CO2 capture processes."
In this new internationally collaborative study, researchers have put forward a solution to this challenge in the form of mixed matrix membranes (MMMs). MMMs are made of two classes of cutting-edge microporous materials, polymers of intrinsic microporosity (PIMs) and metal-organic frameworks (MOFs), thereby achieving enhanced selectivity without sacrificing permeability.
The researchers are developing next-generation polymer membranes based on microporous materials, such as polymers of intrinsic microporosity (PIMs). PIMs are a new class of high permeability polymers invented by UK scientists Profs Peter Budd and Neil McKeown about a decade ago. Now, scientists have incorporated amine-functionalized MOF nanoparticles as nanofillers into PIM polymers, forming new composite membranes that become as selective for separation carbon dioxide as conventional polymer membranes, but allow carbon dioxide to pass through it a few hundred times faster. The combination of high gas permeability and selectivity of these mixed matrix membranes pushes the overall performance to a level that makes CO2 capture economically feasible.
The research team attribute this remarkably improved performance to the interactions between the PIM polymer and the MOF nanofillers. The amine functional groups on MOF nanoparticle surfaces are able to form hydrogen bonding with the rigid PIM polymer chains, therefore allowing polymer chains become physically crosslinked and even more rigid. Such physical interactions tune the size and connectivity of molecular-sized cavities in the membranes and improve their ability to selectively separate molecules, allowing small molecules to pass through while effectively blocking large ones.
Scientists believe these highly permeable and selective membranes provide more energy-efficient separation of carbon dioxide from flue gas emitted from combustion of fossil fuels, potentially reducing the cost of capturing carbon dioxide significantly, and revolutionising CO2 capture technology.
“The new design of mixed matrix membranes made of PIMs and functionalized MOFs opens the possibility of preparation of high-performance membranes from a large family of PIM polymers we are developing, which would enable large-scale, energy-efficient and environmentally friendly gas separation applications more technologically feasible in major global energy processes” said Professor Neil McKeown from the University of Edinburgh, one of the inventors of PIMs materials who was not involved in this study.
Dr Qilei Song, Lecturer in the Department of Chemical Engineering at Imperial College London and a co-author of the paper, said: "These microporous polymer membranes are multifunctional."
"They are not only useful as filters for separation CO2 and other greenhouse gases, but also can be used in many other molecular-level separations, such as air separation for oxygen enrichment, natural gas and hydrogen purification for sustainable energy production, capture of harmful gases from the environment, water purification and desalination, and separation of chemical mixtures in organic solvents, which are actively being studied in the newly-established Barrer Centre at Imperial College London."
The paper "Enhanced selectivity in mixed matrix membranes for CO2 capture through efficient dispersion of amine-functionalised MOF nanoparticles" appeared on June 5, 2017 in Nature Energy. DOI: 10.1038/nenergy.2017.86
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Ms Genevieve Timmins
Academic Services