Apr 2019 - Article in ACS Applied Materials and Interfaces Published

ACS Applied Materials and Interfaces
Alex Cryer's paper on the use of afatinib-functionalised gold nanoparticles (NPs) for treatment of non-small cell lung cancer (NSCLC) is published.
Alexander M. Cryer, Cheuk Chan, Anastasia Eftychidou, Christy Maksoudian, Mohan Mahesh, Teresa D. Tetley, Alan C. Spivey and Andrew J. Thorley, ‘Tyrosine Kinase Inhibitor Gold Nanoconjugates for the Treatment of Nonsmall Cell Lung Cancer’, ACS Applied Materials and Interfaces, 2019, 11, 16336-16346. DOI: 10.1021/acsami.9b02986
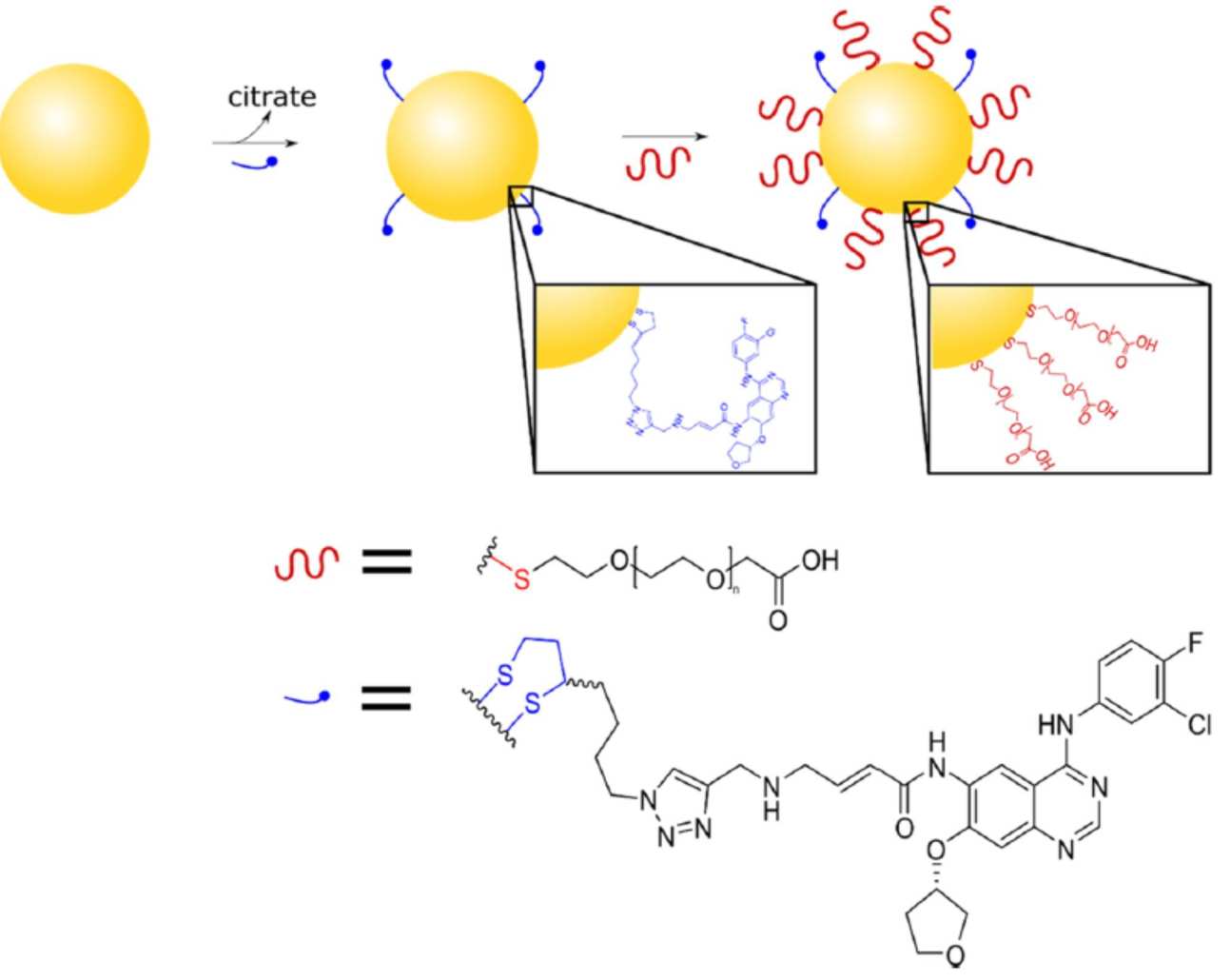
The chemistry in this paper was largely carried out by three successive Imperial College MRes students as part of their dissertation research projects: Christy Maksoudian (2016), Anastasia Eftychidou (2017) and Cheuk Chan (2018) under the supervision of Prof. Alan Spivey and Mr Alex Cryer (a PhD student of Dr Andrew Thorley and Prof Terry Tetley in the NHLI, Imperial College London).
Gold nanoparticles (AuNPs) have emerged as promising drug delivery candidates that can be leveraged for cancer therapy. Lung cancer (LC) is a heterogeneous disease that imposes a significant burden on society, with an unmet need for new therapies. Chemotherapeutic drugs such as afatinib (Afb), which is clinically approved for the treatment of epidermal growth factor receptor positive LC, is hydrophobic and has low bioavailability leading to spread around the body, causing severe side effects. Herein, we present a novel afatinib-AuNP formulation termed Afb-AuNPs, with the aim of improving drug efficacy and biocompatibility. This was achieved by synthesis of an alkyne-bearing Afb derivative and reaction with azide-functionalized lipoic acid using copper-catalyzed click chemistry, then conjugation to AuNPs via alkylthiol−gold bond formation. The Afb-AuNPs were found to possess up to 3.7-fold increased potency when administered to LC cells in vitro and were capable of significantly inhibiting cancer cell proliferation, as assessed by MTT assay and electric cell−substrate impedance sensing, respectively. Furthermore, when exposed to Afb-AuNPs, human alveolar epithelial type I-like cells, a model of the healthy lung epithelium, maintained viability and were found to release less pro-inflammatory cytokines when compared to free drug, demonstrating the biocompatibility of our formulation. This study provides a new platform for the development of nontraditional AuNP conjugates which can be applied to other molecules of therapeutic or diagnostic utility, with potential to be combined with photothermal therapy in other cancers.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Professor Alan C Spivey
Department of Chemistry