Publication in ACS Chemical Biology
by Anna Barnard
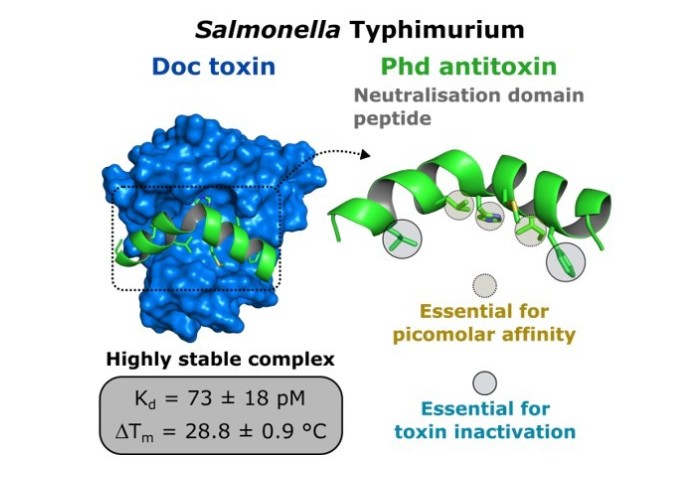
Our recent work on characterisating a toxin-antitoxin pair is now online
Our recent work on 'Characterisation of the key determinants of Phd antitoxin mediated Doc tocin inactivation in Salmonella is now published online in ACS Chemical Biology.
In the search for novel antimicrobial therapeutics, toxin-antitoxin (TA) modules are promising yet underexplored targets for overcoming antibiotic failure. The bacterial toxin Doc has been associated with the persistence of Salmonella in macrophages, enabling its survival upon antibiotic exposure. After developing a novel method to produce the recombinant toxin, we have used antitoxin-mimicking peptides to thoroughly investigate the mechanism by which its cognate antitoxin Phd neutralises the activity of Doc. We reveal insights into the molecular detail of the Phd-Doc relationship and discriminate antitoxin residues that stabilise the TA complex from those essential for inhibiting the activity of the toxin. Co-expression of Doc and antitoxin peptides in Salmonella was able to counteract the activity of the toxin, confirming our in vitro results with equivalent sequences. Our findings provide key principles for the development of chemical tools to study and therapeutically interrogate this important class of protein-protein interactions.
This work was carried out by Dr Dennis Worm and Dr Guilherme Vieira de Castro from the group with input from former group member Dr Fiona Rowan and undergraduate project students Lucy Haggerty, Ana Losada de la Lastra and Oana Popescu. The microbiology work was performed by our collaborators at Harvard Medical School Dr Sophie Helaine and Dr Grzegorz Grabe. Congratulations to everyone involved!
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Anna Barnard
Department of Chemistry