New polymer-based batteries could offer promise for sustainable energy storage
by Sara West

Dr Anqi Wang showing the polymer battery
A team at Imperial College London have developed organic electrode materials which could provide the solution to sustainable energy storage.
Electrochemical energy storage is crucial to the success of Net Zero strategies which aim to reduce greenhouse gas emissions and increase clean energy outputs. There are growing concerns about the high cost, toxicity, environmental pollution, and end-of-life recycling of lithium-ion batteries, which currently dominate the market of portable electronics and electric vehicles. Sustainable organic electrode materials hold the potential to replace these conventional inorganic materials, but they have previously been limited by performance challenges.
Now, an international team led by Dr Qilei Song at the Department of Chemical Engineering of Imperial College London has developed a new type of organic electrode material with promising performance in Li-ion batteries. The work is published in the Journal of the American Chemical Society.
Polymer electrode materials
Conventional lithium-ion batteries typically use inorganic electrode materials such as lithium cobalt oxide (LiCoO2) and lithium manganese oxide (LiMn2O4) which are damaging to the environment and limited in resource.
"Our molecular design approach is a synergistic combination of organic electrode materials and porous polymers developed in recent decades.” Dr Anqi Wang Department of Chemical Engineering
Dr Song’s team have developed organic electrode materials which integrate redox-active organic molecules, which release and store energy, into long-chain polymers. The resulting polymer particles are dissolved and mixed with carbon additives to make battery electrodes.
This newly designed polymer electrode material has improved stability and addresses existing problems with organic electrode molecules, including the loss of storage capacity over time, and slow ion transport and electron transfer – the critical aspect responsible for energy deployment and charging in batteries.
Lead author Dr Anqi Wang said: “To solve the scientific challenges associated with organic electrode materials, we developed new polymer electrode materials that combine solution-processability, redox activity, and sub-nanometre pores. Our molecular design approach is a synergistic combination of organic electrode materials and porous polymers developed in recent decades.”
Nanoscale porosity and solution processability
The polymer electrode materials possess intrinsic sub-nanometer pores that enable fast Li-ion transport during battery operation. The generation of these sub-nanopores is a direct consequence of their unique macromolecular chain structures that are highly rigid and awkwardly shaped.
The other unique property of these polymers is their solution processability, which means they can be dissolved into an organic solvent and then processed into any forms and shapes as a composite or a stand-alone material. This adds greater versatility and simplicity to their use as battery electrodes.

The solution processing method which the team developed, where they dissolved the polymers in solvent and mixed it with electron-conductive carbon additives, also showed significantly improved mixing and contact between polymers and carbon additives including carbon nanotubes.
Such control over interfaces could promote homogeneous current distributions within the electrode, something which has been difficult to achieve in conventional electrode materials, but which is key to ensuring the long lifetime of the battery.
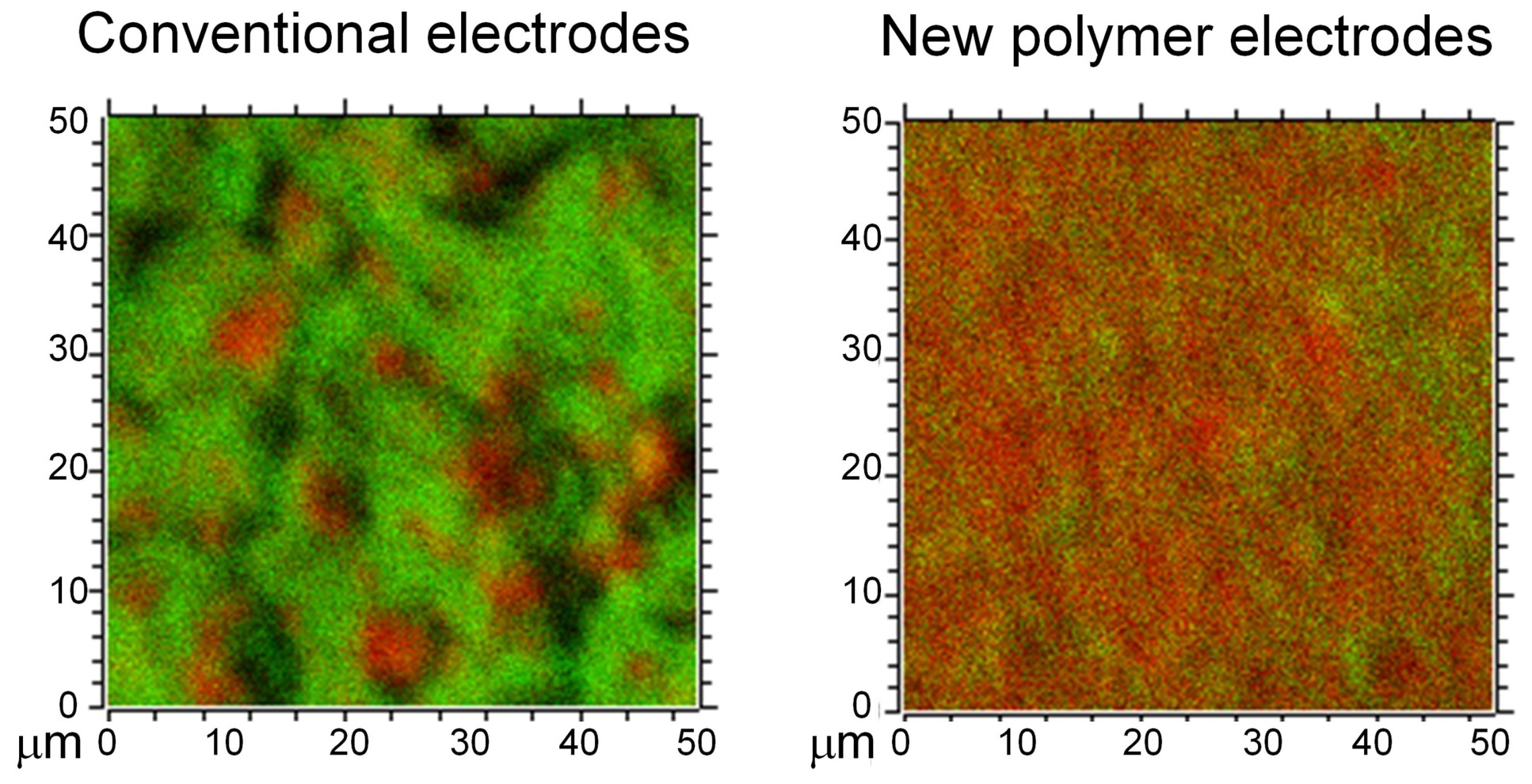
The newly developed battery electrodes demonstrated stable cycling performance without any apparent capacity decay over more than a thousand cycles of charging and discharging.
Co-lead author Dr Rui Tan, who helped develop the battery electrodes, said: “The solution processing technique is suitable for integration with the existing battery electrode manufacturing processes already used in industry at large scale”.
Next steps
Next, the researchers will apply the latest machine learning techniques to screen a large database of organic building blocks to develop the next generation of redox electrode materials with fast conduction of both metal ions and electrons for fast charging organic batteries with long lifespans, and realise the design experimentally through an economically favourable synthetic way.
"Ultimately, we aim to develop low-cost energy storage technologies to satisfy the ever-growing demand of electrification of many sectors and to facilitate the integration of renewable energies.” Dr Qilei Song Department of Chemical Engineering
The molecular design approach is also applicable to materials for other energy storage systems, such as sodium-ion (Na-ion) batteries and redox flow batteries. The team is seeking opportunities to collaborate with other research teams to explore these applications, and to work with Faraday Institution to investigate the degradation and manufacturing of these polymer electrode materials.
Dr Qilei Song, corresponding author of the paper, said: “Organic electrode materials have great potential for use in a variety of electrochemical devices for energy storage applications. Ultimately, we aim to develop low-cost energy storage technologies to satisfy the ever-growing demand of electrification of many sectors and to facilitate the integration of renewable energies.”
The molecular design of these polymers was carried out in collaboration with Professor Neil McKeown at University of Edinburgh and Professor Kim Jelfs in Department of Chemistry at Imperial College London. This work also involves collaborations with researchers from University College London, Nankai University, and Tongji University. This research was funded by Engineering and Physical Sciences Research Council (EPSRC), part of the UKRI, European Research Council, and Royal Society.
-
Solution-processable redox-active polymers of intrinsic microporosity for electrochemical energy storage by Anqi Wang et al., published 8th September 2022 in Journal of the American Chemical Society.
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Sara West
Communications Division