Genetic Causes of Three Previously Unexplained Rare Diseases Identified

A new computational approach has helped researchers identify previously unknown genetic causes for three diseases.
Researchers led by Dr Graeme Birdsey at the National Heart and Lung Institute and Professor Pia Ostergaard at St George’s, University of London have collaborated to uncover the implications of a loss-of-function in the ERG gene has for the onset of primary lymphoedema. Lymphoedema is a long-term (chronic) condition that causes swelling in the body's tissues. The work was carried out with colleagues at the Icahn School of Medicine at Mount Sinai in New York who have developed a computational approach to screen whole genome sequencing data for rare disease-causing genetic variants.
"Our ongoing studies will help widen an understanding of the regulation of genes involved in the onset of primary lymphatic diseases" Dr Graeme Birdsey
Rare diseases affect approximately 1 in 20 people, but only a minority of patients receive a genetic diagnosis. Fewer than half of the 10,000 recorded rare diseases have a known genetic cause. Genome sequencing of large cohorts of rare disease patients provides a route towards discovering the genetic causes that remain unknown. One major endeavour, the 100,000 Genomes Project, sequenced the genomes and collected clinical phenotype data for 77,539 participants. However, large genetic datasets are challenging to work with, slowing down research significantly. To overcome this, investigators at Mount Sinai developed a statistical method that focused only on rare genetic variants, enabling them to perform analyses using a small database (a `Rareservoir’), only 5.5GB in size, hastening progress significantly.

Using the new computational approach, relationships between 20,000 genes and 269 rare diseases were analysed by the Mount Sinai team. They identified 260 associations between genes and rare disease classes, including 19 associations previously unrecorded. Through an international collaboration, the authors validated novel associations by identifying additional cases in other countries and through experimental and bioinformatic approaches. This has led to the discovery of previously unknown genetic causes of three rare conditions: primary lymphoedema, thoracic aortic aneurysm disease (a swelling or bulging at any point along the aorta), and congenital deafness. The work was done in collaboration with colleagues at the University of Bristol, UK; KU Leuven, Belgium; the University of Tokyo; the University of Maryland; Imperial College London, and others from around the world.
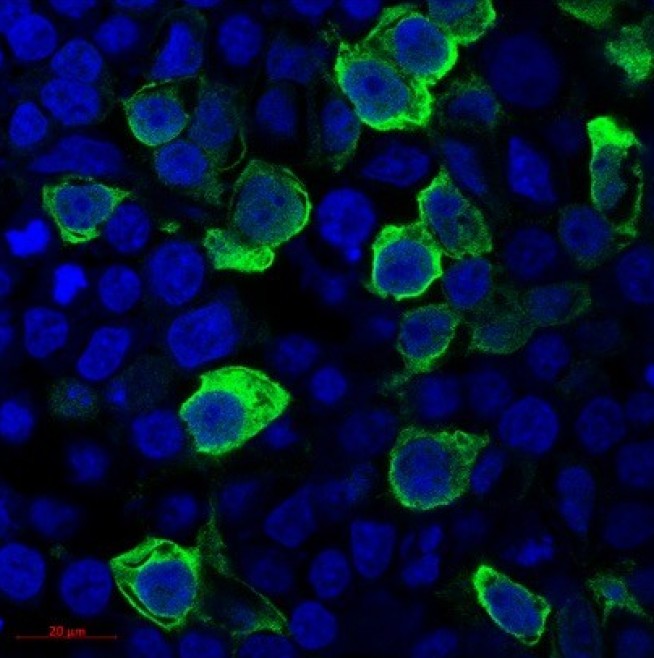
Lymphoedema is a chronically debilitating disease that affects about 120,000 people in the UK, of which 28,000 are estimated to have primary lymphoedema. Primary lymphoedema is an inherited genetic condition caused by abnormal development of lymphatic vessels or failure of lymphatic function, due to mutations in key genes. Approximately 40% of primary lymphoedema cases are due to identifiable genetic causes, however, there remain numerous patients whose genetic causes are unknown. Dr Graeme Birdsey, Dr Daniela Pirri and Dr Karen Frudd at NHLI worked closely with colleagues at St. George’s, University of London to uncover the role of loss-of-function variants in the ETS-family transcription factor ERG. By overexpressing ERG in a cell line, the Imperial researchers were able to demonstrate that the ERG variants identified in the primary lymphoedema patients could not enter the cell nucleus and remained in the cytosol, preventing ERG from binding to DNA and carrying out its function as a transcription factor. This is the first reported association between cardiovascular disease and patients with genetic variants in ERG, so could have a large impact on future treatments. Ongoing studies by the Birdsey group are uncovering the molecular mechanisms through which these and other genetic variants of ERG cause the dysfunctional vasculature associated with primary lymphoedema.
In this study, the authors only considered certain types of genetic variants called single nucleotide variants and short insertions or deletions in genes that code for proteins. Future work will explore different types of variants and genes in future. The Rareservoir will be an invaluable tool to hasten discovery and replication across the different large genetic datasets that now exist across the globe.
The paper “Genetic association analysis of 77,539 genomes reveals rare disease etiologies” is published in Nature Medicine.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Ms Helen Johnson
Communications Division