Cells develop a ‘thicker skin’ under extreme gravity
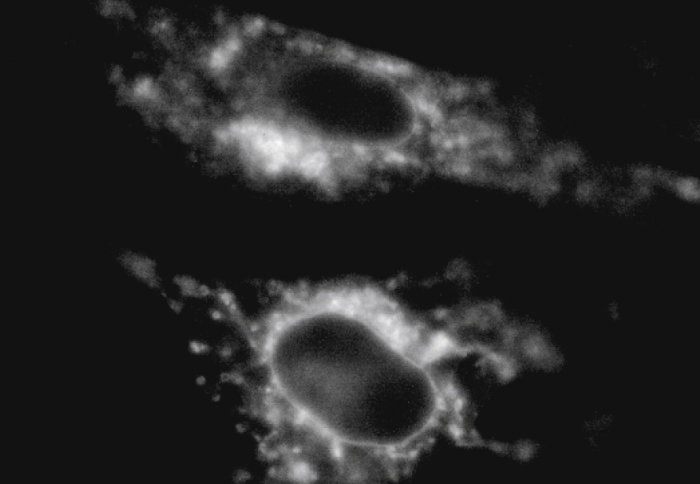
A high-gravity experiment has revealed how cells keep their shape under pressure.
Scientists have probed how cells respond to high gravity with a technique that could transform how we look at cellular life.
Using the European Space Agency’s (ESA) Large Diameter Centrifuge in the Netherlands, Imperial and ESA researchers spun cells at high speeds that simulate a gravitational force 15 times stronger than on Earth (15g). As gravity grew higher, the viscosity of cell membranes increased, similar to developing a ‘thicker skin’ to hold its shape under pressure.
Actually being able to conduct detailed microscopy experiments inside of a centrifuge which is spinning at 15g is not very easy. Dr Nick Brooks
This is the first study of cell membrane viscosity – the measure of how sticky or fluid the cell walls are – under extreme gravity. This study brings researchers closer to working out how cells function and how certain diseases affect cells. Viscosity controls how quickly key reactions occur or whether they do at all, while diseases like diabetes and Alzheimer’s can change the viscosity within cells.
Dr Nick Brooks, co-author and Senior Lecturer in the Department of Chemistry, explains: “Studying how cells adapt to extreme gravity helps us understand how cells can resist environmental stress in general – from viral attacks and chemical imbalance to extreme cold and extreme heat.”
The study was also a technical feat, he says: “Another really exciting thing was showing we could do it. Actually being able to conduct detailed microscopy experiments inside of a centrifuge which is spinning at 15g is not very easy.”
Hypergravity environment
This multidisciplinary project brought together chemists, physicists and ESA scientists. The study was led by Imperial PhD graduate Emma Woodcock as part of the Spin Your Thesis! competition that gives students access to the ESA’s centrifuge. The centrifuge has four arms that span its eight-metre diameter.
At full speed, the containers at the end of the arms move at 101km/hour. Inside of each container is an experiment that can simulate a hypergravity environment up to 20g. To put that in perspective, some of the most powerful rollercoasters can reach 5g for a few seconds.
Beyond the logistical challenges of securing a camera and microscope in the container, researchers had to pioneer a new technique that could withstand extreme gravity. Associate Professor in the Department of Chemistry Dr Marina Kuimova developed molecules that light up as viscosity increases in the cell membrane.
The molecules have a spinning axis that rotates more quickly in low viscosity and moves more slowly as the membrane stiffens. When the molecule is not spinning, it emits bright fluorescence that researchers can measure quantitatively. This method is also promising because it doesn’t interfere with the chemical make-up of the cell as drug molecules can.
Dr Kuimova explains: “This technique opens a whole new way of looking at how cells work. We usually monitor smaller changes in viscosity under normal physiological conditions, but it was exciting to apply our technique to conditions when the cells were pushed to the extreme.”
Dealing with pressure changes
Researchers tested the effect of high gravity on two types of cells: general mouse cells and specialised human cells that line blood vessels. They saw the greatest and fastest change in the human cells.
These cells often have to deal with changes in pressure from sharp corners in vessels, plaque build-up and increased speed of blood flow. This is particularly interesting as it illustrates what cells might be subjected to with arterial diseases, though on a larger scale.
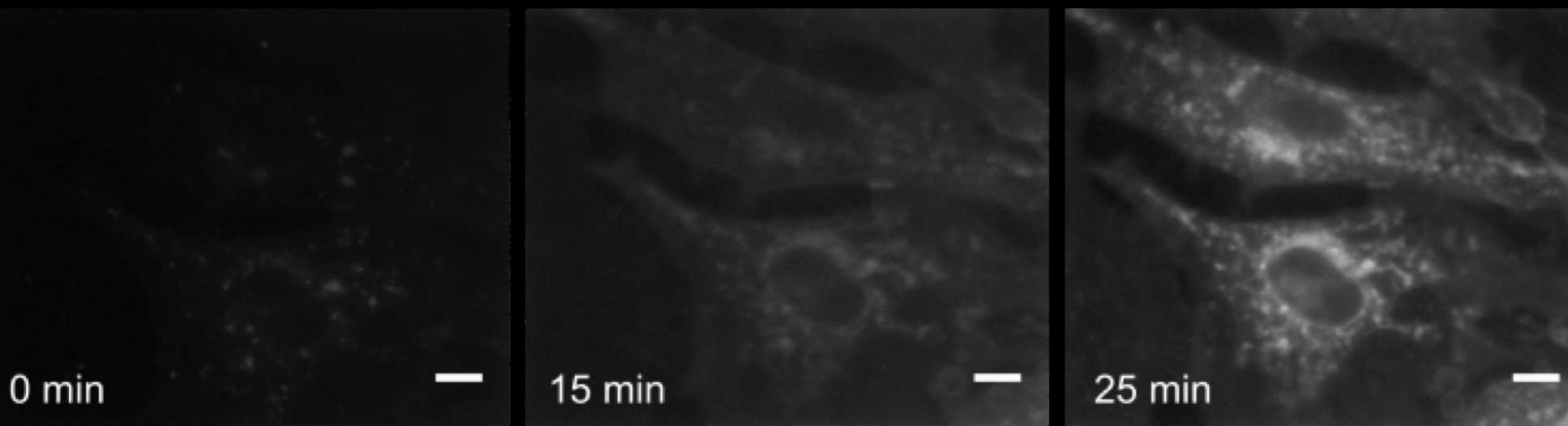
Both types of cells had very fast response in stiffness to a gravity ramp. This means that it was largely a physical response of membrane components to the increased pressure rather than the cell sensing the change and setting off its biological machinery to produce a mechanical change.
Going forward, researchers can look at how the cell tells the membrane to increase its viscosity. This approach could also be used to study the viscosity of different parts of the cell, how this is implicated in diseases and how cell membranes control other things.
-
‘Measuring Intracellular Viscosity in Conditions of Hypergravity’ by Emma Woodcock, Paul Girvan, Julia Eckert, Ismael Lopez-Duarte, Marketa Kubankova, Jack van Loon, Nicholas Brooks and Marina Kuimova is published in the Biophysical Journal.
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Juanita Bawagan
Centre for Languages, Culture and Communication