‘Vaccines have saved more lives than any other medicines’- Robin Shattock
by Ryan O'Hare

Prof. Robin Shattock on BBC Question Time
A top vaccine expert has tackled public concerns that a rapid rollout of COVID immunisation programme would be unsafe.
Professor Robin Shattock, from the Department of Infectious Disease, said that vaccines have saved “more lives than any other medicines” and that the benefits far outweigh the risks.
The immunologist, who leads Imperial’s COVID-19 vaccine research, made the comments on the BBC’s Question Time last night, appearing on a panel alongside Health Secretary Matt Hancock, and which focused heavily on the Government’s plans to rollout millions of doses of Pfizer-BioNTech’s COVID-19 vaccine candidate.
“I think we need to remember that vaccines, next to clean water, have been the single most effective public health implementation globally. They have saved more lives than any other medicines,” said Professor Shattock.
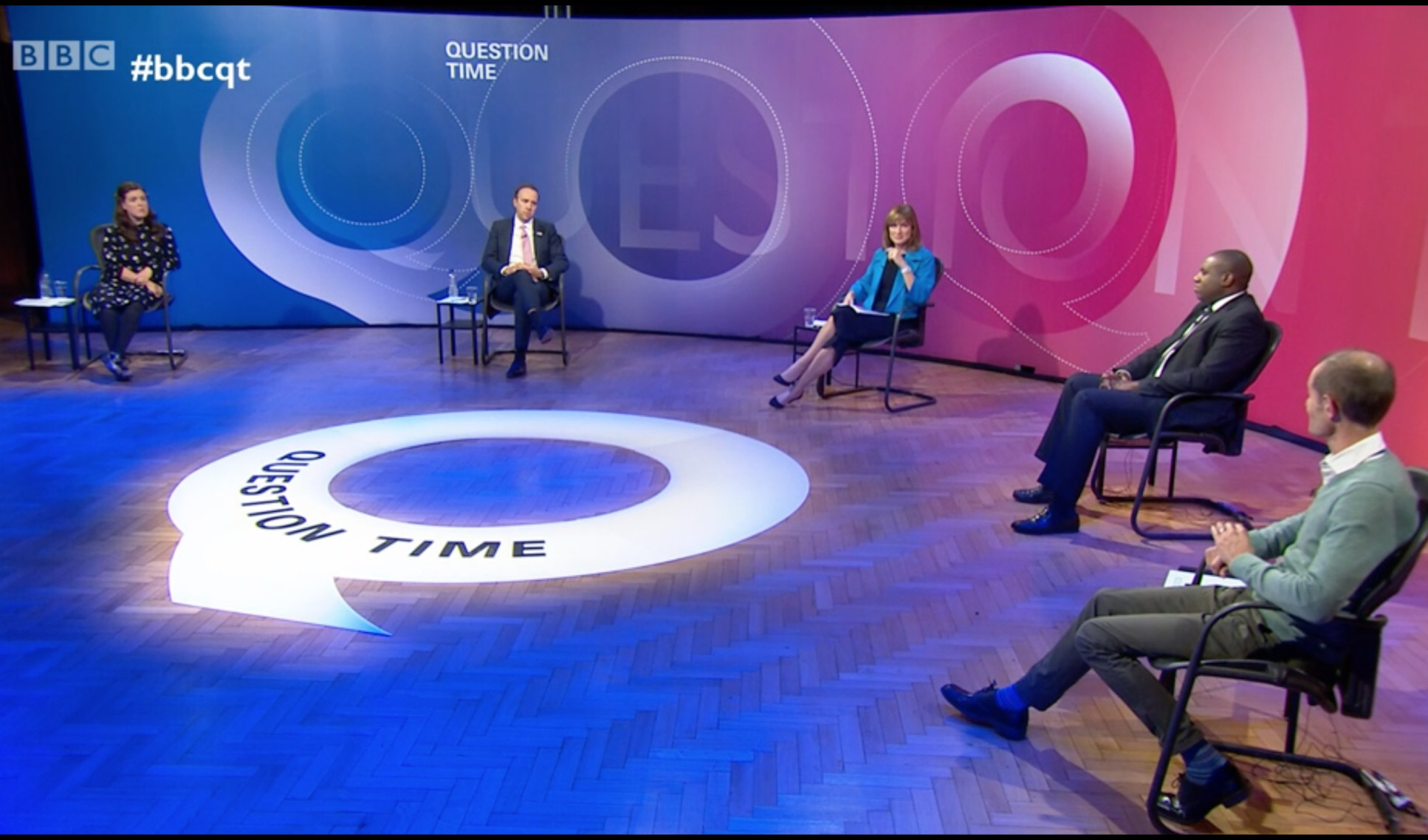
Responding to issues of public confidence in vaccines and the potential for side effects from immunisation against COVID-19, he explained: “You’re getting an aspect of viral infection that’s so much [more] minor than the real virus…the risk of having a severe outcome from COVID-19 far outweighs any theoretical risk of a vaccine.”
Immense challenge
This week the UK Government announced the purchase of 40 million doses of the Pfizer-BioNTech vaccine, an RNA-based candidate requiring two doses. It provided interim guidance on which groups would be prioritised to receive the limited supply.
“The logistical problem is immense,” said Professor Shattock, commenting on the issues of rolling out a COVID-19 vaccine nationally and the need to prioritise access to high-risk groups.
“There’s about 10 million [people in the] vulnerable population, and if you started vaccinating in January – given that it takes five to six weeks to get immunity – and you did a million people a week, we wouldn’t get that 10 million vaccinated until well into April.”
One of the major concerns raised around the Pfizer-BioNtech vaccine is the low temperatures at which it needs to be kept (between -70C and -80C) and the logistical challenges of cold-chain distribution to get the millions of doses to where they are needed.
Other vaccines, including Imperial’s self-amplifying RNA vaccine candidate, could help to overcome these logistical issues due to being more stable at higher temperatures (closer to 0C).
"We are already looking at how to make our vaccine stable at refrigerated temperatures, and it is absolutely possible," Professor Shattock told The Telegraph.
Lasting legacy
Highlighting the benefits of the self-amplifying RNA platform behind Imperial’s COVID-19 vaccine candidate, he told The Independent: “This is a completely new technology. If things go well, we can help to create an important legacy for the UK.”
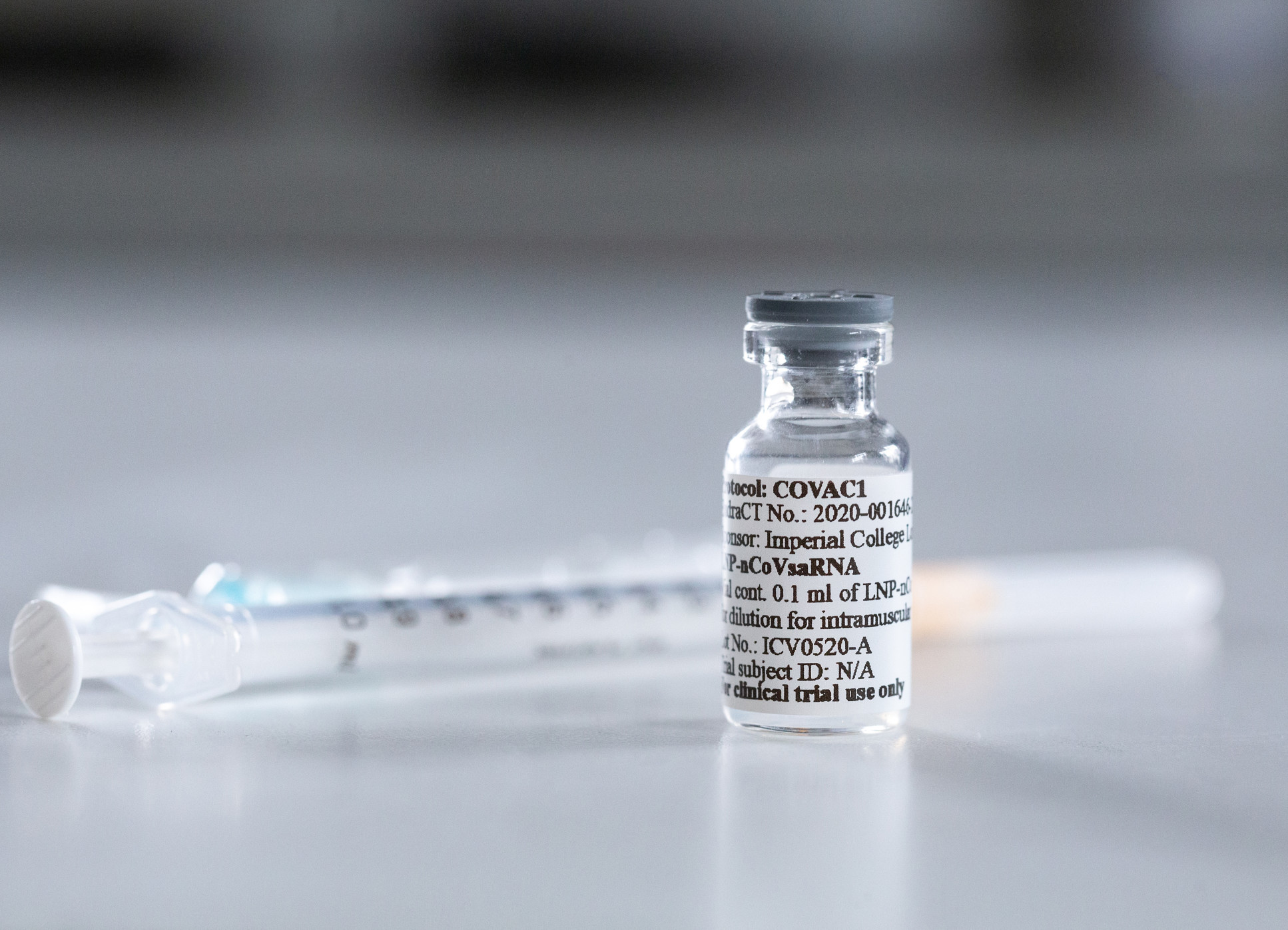
Professor Shattock’s team at Imperial is currently one of two UK-led efforts to develop a COVID-19 vaccine (alongside Oxford-AstraZeneca), a point he hammered home on Question Time.
“We really need to ramp up UK manufacturing capacity so that we can make vaccines, and we have cutting edge technology in this country,” explained Professor Shattock.
“From a pragmatic point of view, the government has purchased six vaccines, five of those are from overseas, and that’s increasing our reliance on overseas production.
“We need to think in terms of the strategy and how we are going to nurture a vaccine industry, which is more than just start-up, that’s sustainable and has a long-term strategic plan.”
See the Imperial COVID-19 Vaccine Trial and COVID-19 Response Fund webpages for further information.
-
Watch the full episode of Question Time on the BBC iPlayer
Read the full interview in The Independent
Read the full piece in The Telegraph
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Ryan O'Hare
Communications Division