
Imperial news
In this section
From the outset of the pandemic, Professor Robin Shattock and colleagues in the Department of Infectious Disease have worked to develop a safe and effective COVID-19 vaccine using their innovative, self-amplifying RNA technology.
While traditional vaccines are often based on a weakened form of a virus, the Imperial team took a new approach to developing their COVID-19 vaccine candidate, which instead uses parts of the virus’ genetic code called self-amplifying RNA (or saRNA). Once injected into muscle, cells rapidly produce copies of a protein found on the outside of the virus. This trains the immune system to respond to the coronavirus so the body can easily recognise it as a threat in future.
The results from the first clinical trial showed no short-term safety concerns and indicate that the vaccine can generate immune responses against COVID-19 in up to 87% of people, even at extremely low dose levels.
In 2020, Professor Shattock also founded the startup VaxEquity with Morningside Ventures to develop transformative RNA therapeutics using Imperial’s next-generation saRNA platform. In September 2021, VaxEquity announced a strategic, long-term research collaboration with biopharmaceutical company AstraZeneca. This partnership will build on the success of Imperial’s response to the pandemic and further explore how the cutting edge saRNA technology can be optimised and applied across various therapy areas.
Page last updated: 8 July 2022
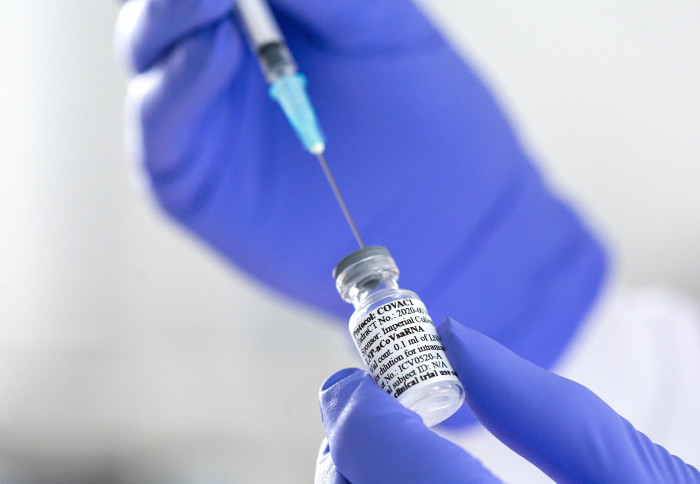
The purpose of the clinical trial (COVAC1) was to evaluate whether Imperial's new self-amplifying ribonucleic acid (RNA) COVID-19 vaccine candidate is safe and whether it produces immune responses against the coronavirus, SARS-CoV-2.
Phase I was the first of multiple steps in the clinical trial and focussed on evaluating the safety of the vaccine and finding the right dose to give.
Different dose levels of the saRNA COVID-19 vaccine were been tested for safety, and the ability to produce an immune response in volunteers.
The study team started with a very low dose (0.1 micrograms) and gradually progressed through the different dose levels up to 10 micrograms. In total, the Phase I study enrolled 192 adult volunteers in the low dose and higher dose cohorts.
The results of the Phase I study, published in the journal eClinicalMedicine, and reported on the Imperial College news site, suggest that the vaccine can generate immune responses against COVID-19 in up to 87% of people, even at extremely low dose levels – the lowest of any COVID-19 vaccine candidate worldwide.
Coronaviruses are covered in spike proteins that stick out from their surface and which the virus uses to get into cells. Once inside, the viruses can produce thousands/millions of copies of themselves and can then spread to infect other cells.
When the outbreak first emerged, scientists in China isolated samples of the SARS CoV-2 virus from patients with COVID-19 and were able to sequence its genetic code – which is made up of Ribonucleic Acid (RNA).
The Imperial team, led by Professor Robin Shattock, focused on the part of this sequence that holds the blueprint for the spike protein. They were able to recreate the sequence using enzymes in the lab and generate copies of the RNA without the need for animal cells or human stem cells.
The result is short strands of self-amplifying RNA which hold all the information needed to make the spike protein. These strands are packaged into tiny fat droplets, which form the final vaccine.
 When the Imperial vaccine is injected (into the arm, or the leg) the cells of the muscle will take up the tiny fat droplets and the RNA they contain.
When the Imperial vaccine is injected (into the arm, or the leg) the cells of the muscle will take up the tiny fat droplets and the RNA they contain.
Once inside the cell, the self-amplifying RNA produces copies itself, which can instruct the cell’s own machinery to make the coronavirus protein. This process takes place in the cytoplasm of the cell and so doesn’t affect or change the cell’s own genetic material (its DNA).
The muscle cells will then produce lots of the spike protein – but not the whole virus. Some of the proteins will be presented on the surface of the muscle cells, as part of the normal process the body uses to show the immune system what’s going on inside cells.
When the immune system comes across these tiny spikes, it recognises them as foreign and creates antibodies. These antibodies have an important role in fighting viruses as they bind to the spike proteins and may prevent the virus from infecting cells. This process also creates a lasting memory of the spikes so that the body ever comes across them again, it will recognise them and be able to mount a quick response.
Since the outbreak at the end of 2019, the novel coronavirus SARS CoV-2 has spread rapidly around the world and caused hundreds of thousands of deaths. One of the reasons it has been so harmful is that it is a new or novel virus, and people had no immunity against it.
Ultimately, the aim of the vaccine is to trick the body into thinking it has already seen the virus and made an immune response, so when you come into contact with it in real life, you should already be immune.
The Imperial COVID-19 vaccine trial was made possible thanks to a number of generous research awards and donations.
We are sincerely grateful to everyone who has donated to support Professor Shattock’s vaccine work. These donations, at all levels, have enabled the research team to move rapidly with the development and testing of the vaccine. More than £4.5 million has been donated to support Professor Shattock’s vaccine work, including:
Other donors have chosen to give anonymously. These donations come as part of a groundswell of support from the College’s alumni and donor community, which has seen hundreds pledge a gift to support Imperial’s COVID-19 response.
To discuss a gift to support COVID-19 vaccine research at Imperial College London, please contact Angela Bowen (Director of Development, Faculty of Medicine) on +44 (0)20 7594 1277 or email angela.bowen@imperial.ac.uk

Imperial news

Imperial news
Health
Health

Imperial news
Imperial news

Health

Imperial news
Health
Health

Health

Imperial news