Nature is trying to kill you, but science has your back
by Ryan O'Hare

A new book explores disease-causing microbes, the tools we’ve developed to fight them, and the scientists who’ve advanced infectious disease research.
Pus-filled boils, weeping lesions, and projectile vomiting; just a few of the delights brought on by any number of pathogens determined to snuff us out.
But while infectious diseases are ever present, centuries of scientific progress have helped us to wage war on the malady-causing microbes at their heart – ensuring some of the nastiest pathogens, like the smallpox virus, are resigned to the pages of medical textbooks.
In his new book Infectious: Pathogens and how we fight them Dr John Tregoning, from Imperial’s Department of Infectious Disease, delves into the history of infectious disease research, from the ‘ologies’ at the bedrock of research into pathogens, to the rapid development and deployment of therapies and vaccines to keep them at bay.
Ryan O’Hare spoke to Dr Tregoning to find out more about the book and some of the unsung heroes who have helped us to better understand bacteria, viruses and other pathogens, giving us a crucial leg up in our fight against infectious diseases.
Listen to the full interview, recorded in October 2021. Abridged answers below edited for style and clarity.
Q – What can readers expect from the book – is this a COVID-19 book that will give us new insights into the pandemic? Or should readers expect something else?
John Tregoning: So it's not a COVID-19 book, it's a kind of product of the year because I wrote it during lockdown, but it's actually a much broader science history of infectious diseases.
The first part is about the science behind infectious diseases: how our body fights them off, how we understand about viruses and bacteria. And then the second half is about the therapies: the drugs and the vaccines that we used to prevent and to treat infectious diseases.
Q – Was there anything that surprised you about your own area of research? Or that might influence your work the next time you're at the lab bench?
JT: The thing that really came out for me is the fascinating history of science. We all know some of the very big names, like Pasteur or Fleming, but actually, there's lots of people who've contributed to science. Unearthing these extra people who've contributed and changed the way we think about science, that's been really enjoyable.
Just thinking about science, it really it is a team effort. It's not about that single person at the top. It's about everyone building together and all those kind of little building blocks that go together to make the big breakthrough. So whatever you're doing scientifically, it does somehow contribute to the greater whole.

Q – You mention science being a team effort, and lots of people who may have gone unrecognized. Is there anyone you would like people to know about, who may have gone unnoticed before?
JT: I think the most important person who possibly people haven't heard of is a scientist called Maurice Hilleman, who worked as a vaccinologist in the United States in the 50s, 60s and 70s.
He developed 40 different vaccines during his career, eight of which we still use today around the world to protect children from diseases. His impact and his kind of legacy of saving people from death and disease is enormous, and yet it's often kind of unknown.
Q – There was also a picture you showed at the book launch, of a scientist giving one of his daughters a vaccine. It was an amazing picture. Who was that?

JT: That's the same man, that's Maurice Hilleman.
So what happened was he'd been working on the mumps vaccine and his older daughter Jeryl Lynn got infected with mumps.
On the way to rushing her to hospital he took some swabs from her throat to the lab at the same time, isolated the mumps virus, and then made an attenuated strain – so a strain that doesn't infect you.
There's an image of his daughter, his younger daughter, getting injected with this vaccine while her older sister watches on, and the younger daughter doesn't look too impressed about it.
Q – The book covers quite a lot of ground. But is there a single advance, breakthrough or pinnacle moment that you think stands out from the whole history of infectious disease?
JT: For me, one of the most staggering things is the change in HIV treatment. I'm in my mid-40s and I remember HIV emerging in the 80s. It was this terrifying disease that was killing people aggressively, untreatably, and it was seen as a sort of death sentence.
But now we have developed drugs – and in part that was work done by people at Imperial into testing these drug combinations – to treat it, which means HIV can now be a long chronic stable condition. Just in the space of my lifetime to go from something that that was definitely going to kill you to something that could be managed with access to drugs is an incredible change.
I guess the other one is the is the speed of the COVID-19 vaccines. I think it's mind-blowing that we went from January last year with a basically unknown pathogen and we were all beginning to think ‘Oh, this could be bad’ – to a sequence, to a vaccine, to a rollout, within less than a year, with technologies that were new and relatively untested in kind of this this scale.
That kind of speed of development has been amazing, and it is kind of the tip of the spear. It's 20 years’ work [that] went into this one year of accelerated development. But just seeing that kind of thing happen within my research career is also really exciting.
Q – Is there anything you think the public gets wrong about vaccines that we really shouldn't - the most important thing that we get wrong?
JT: I think the biggest challenge with vaccines is that they are a preventative therapy, so you are taking something to prevent a disease you may never get. It's not dissimilar to wearing a car seat belt, you know, you wear it and most of the time you don't need it.
I think as diseases get rarer and rarer… so I've never seen anyone get measles or mumps or rubella or whooping cough, you know, there's huge numbers of things that we don't see… then it becomes very hard to then equate the risk of that very nasty disease.
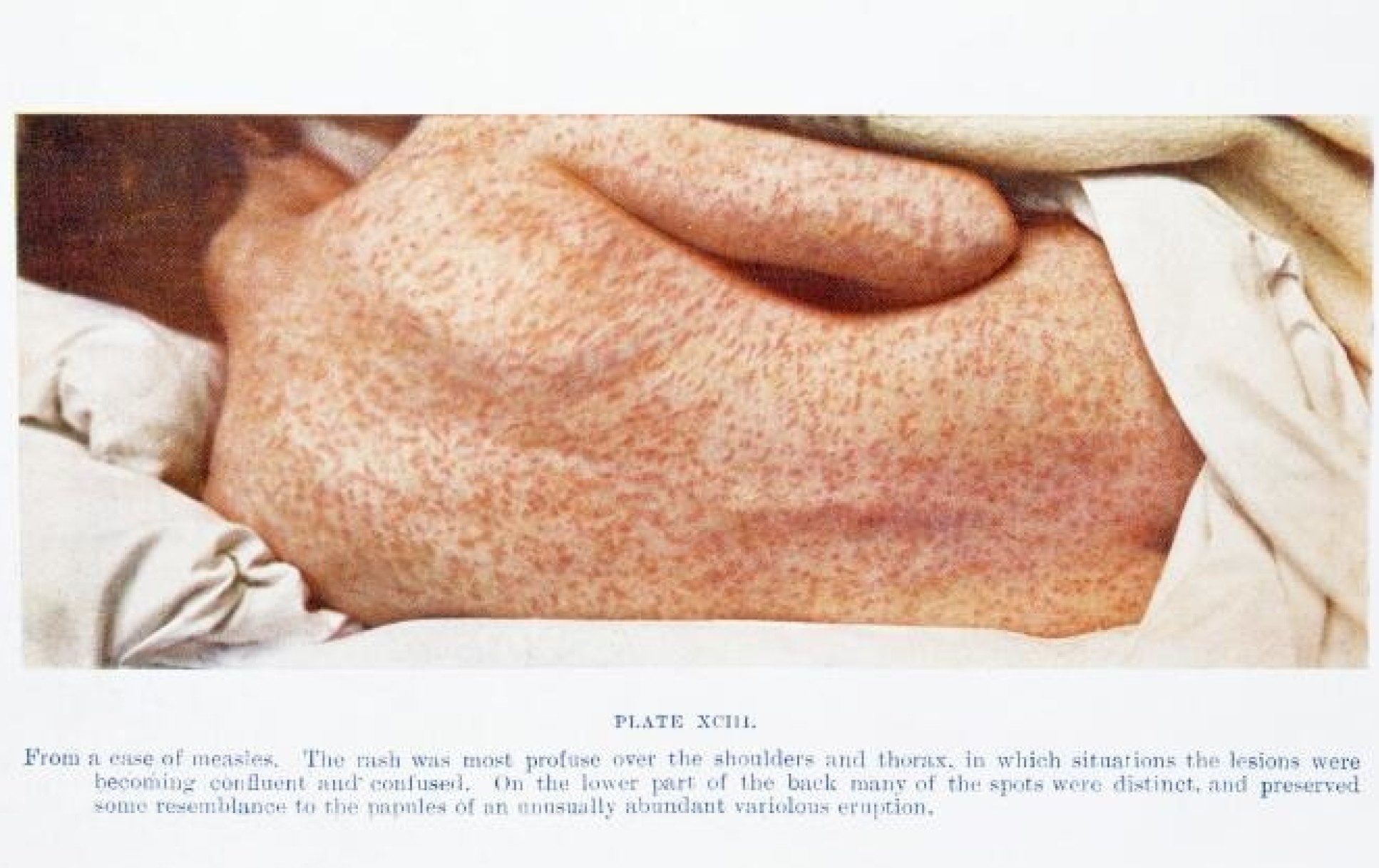
Measles is a catastrophically nasty disease. It has long term disability impact on people who get it. But because we don't see it then the kind of perceived risk of the vaccine gets much bigger than the actual risk of the disease.
So it's that kind of cost/benefit and it's very hard to do if you're not being exposed to the diseases. In countries where [infectious] disease prevalence is much higher, vaccine uptake is much higher, because the immediate benefit is seen.
Q - The book covers a lot of ground, including some of the pioneers of infectious disease research, some of their breakthroughs are a wonderful mix of ingenious and disgusting at the same time. Did you come across like a favourite of yours that was that will stay with you?
JT: Yes, there was a microbiologist, a French-Canadian called Félix d'Hérelle, and he encapsulates an amazing period of 20th century history in terms of how his life played out, as well as being a scientist who discovered something very important.
His scientific breakthrough was to demonstrate that there were viruses that could actually infect bacteria. We think of bacteria as being infectious, but actually there are things that live on top of the bacteria that can kill them.
He was arrested by the German army...set up a failed chocolate factory, and he also got into a scientific duel with another scientist. Dr John Tregoning Imperial College London
He showed you could take a finely filtered extract of chicken poo and put it onto a bacterial culture and kill the bacteria, and within that there were these viruses called bacteriophages. So he was one of the first people to demonstrate that.
But he was doing that in the context of the 1920s and 1930s, when there's this huge kind of global political change.
In his personal life he lived in Russia for a bit, his scientific mentor at the time was having an affair with the head of the Russian secret police, he got in trouble with them. He then was arrested by the Wehrmacht – the German army – during the 1940s.
He set up a failed chocolate factory. And he also got into a scientific duel with another scientist over who actually identified the bacteriophage in the first place. So it's just a sort of crazy colourful life on top of like an important scientific discover as well.
I don't think it actually moved as far as in person, with pistols, but he did write into a scientific journal and challenged another man to a duel. So it's something that maybe we should be bringing back: pipettes at dawn.
Q – At the launch of the book recently at the Royal Institution, you gave two quotes to summarize where we are in the fight against infectious disease which say either ‘the battle is won’ or that ‘the fight is hopeless and it's an arms race we can never win’. On that kind of scale, where do you think we are?
JT: I'm an optimist, you know, if I had to go one way or the other, I would sit towards the battle being won.
I think you or I are much more likely to die of a heart attack or a non-infectious disease than we are of an infectious disease. I think the landscape has completely changed.
There are challenges, there are threats on the horizon. Antibiotic resistance is the biggest, most pressing infectious disease problem. I think there will be other pandemics. As climates change or as human interactions with animals change we're going to see other zoonotic infections emerge.
These things will happen, either acutely or kind of chronically, but we are in such a different place to where we were 100 years ago.
Q - Are there any lessons from the COVID-19 pandemic in terms of infectious disease research and how we view, understand, and deal with pathogens?
JT: I think the biggest lesson for me is that you need the research in the past to then have the emergency push in the future, so it's not a tap that can just suddenly be turned on.
All of the [COVID-19] vaccines came out of 20 years of research. The Oxford [Astra-Zeneca] vaccine was another program that was then repurposed into a SARS vaccine in kind of quite quick time, so the stuff was already there. So you have to have what's known as a warm base. Things have to be running and kind of set up and go.
(Credit: Ash Uruchurtu)
The second thing is you need to have a functioning public health infrastructure. You need the diagnostics. You need that kind of structure. That, unfortunately, has been kind of shrinking in the UK over the last 10= years and I think if it been at the kind of levels it needed to be, maybe the track and trace and the testing might have been quicker going forward.
I think the interface with science and government is very complicated. I think we are lucky that we do have integrated scientific advisors in the government, but maybe there will be a need to be a kind of a bit of a shake up of how that advice is integrated in that and how that moves into policy.
Then going forward, I think we see a space, certainly in my field, for more localized national vaccine production. Historically, there were big hubs where they made all the vaccines, so the [Serum Institute] in India makes a huge amount of vaccines for the world, but that they don't have capacity for this kind of surge capacity. So building newer surge capacity that can be either maintained, or do other things, then that would be important going forward.
Q - If there's one thing that people take away from your book, what would it be?
JT: I think it's the message of optimism. It's that science and ingenuity has led us to being able to deal better with infectious diseases. We are so much better placed to cope with infections than we were 100 years ago. We know what's infecting us. We know how to treat them. We know how to prevent them.
-
Dr John Tregoning is a Reader in Respiratory Infections in the Department of Infectious Disease at Imperial College London.
Infectious: Pathogens and how we fight them by John Tregoning is published by Oneworld Publications.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Ryan O'Hare
Communications Division