Autoimmune diseases and twisting lights: News from the College

Here’s a batch of fresh news and announcements from across Imperial.
From genes that may increase the risk of autoimmune disease, to new twisting laser lights to visualise molecules, here is some quick-read news from across the College.
Neurodegenerative disease dynamics
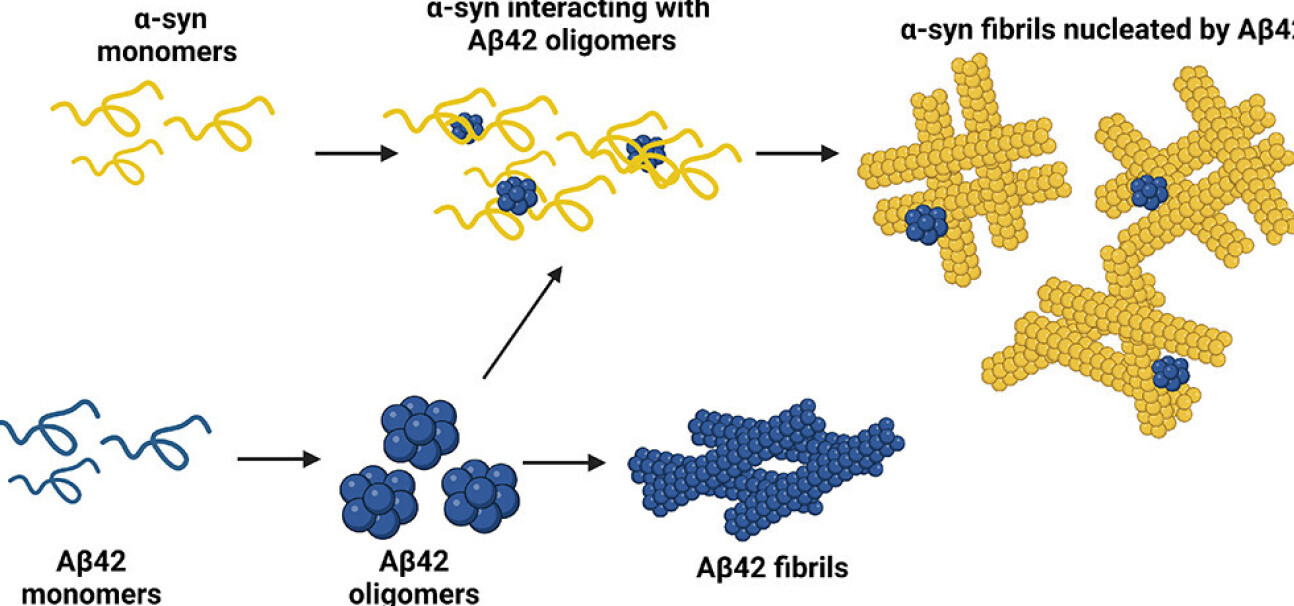 Many neurodegenerative diseases, such as Alzheimer’s (AD) and Parkinson’s (PD) are associated with the accumulation and aggregation of certain proteins that form deposits in the brain. Generally, AD and PD are associated with aggregates of two different proteins: amyloid-β and α-synuclein respectively. Sometimes, both protein aggregates are found in the same patient diagnosed with either disease. How the two proteins interact, and what this means for treatment, is a complex question.
Many neurodegenerative diseases, such as Alzheimer’s (AD) and Parkinson’s (PD) are associated with the accumulation and aggregation of certain proteins that form deposits in the brain. Generally, AD and PD are associated with aggregates of two different proteins: amyloid-β and α-synuclein respectively. Sometimes, both protein aggregates are found in the same patient diagnosed with either disease. How the two proteins interact, and what this means for treatment, is a complex question.
Now, researchers from the Molecular Sciences Research Hub at Imperial and colleagues in Switzerland have uncovered a mechanism where amyloid-β can cause α-synuclein to aggregate. The proteins go through three stages, first existing as monomers (single units), then combining into oligomers (small accumulations) and finally fibrils (rod-like structures that make up the deposits).
The team were able to show that where both proteins are present, the oligomers of amyloid-β, instead of progressing to fibrils, become a point around which fibrils of α-synuclein accumulate, helping them form. This insight could help researchers design new therapies that target the interactions between these proteins and prevent their accumulation.
Read the full paper in the Journal of the American Chemical Society.
Genetics of inflammation
 Researchers from Imperial, working as part of a large international consortium, have found genetic factors which influence the levels of inflammatory proteins in the blood, and link these to the risk of developing autoimmune diseases.
Researchers from Imperial, working as part of a large international consortium, have found genetic factors which influence the levels of inflammatory proteins in the blood, and link these to the risk of developing autoimmune diseases.
The study, which is one of the largest studies to date, looked at the blood of over 15,000 people to find 180 genetic loci that affect the level of 91 proteins in the blood. Researchers found 50 new genetic associations as part of the research that were previously unknown. They then combined this information with genetic studies of autoimmune diseases to reveal proteins that play a causal role in the development of these diseases.
They found a protein called lymphotoxin-alpha may play a role in Multiple Sclerosis (MS). Working with the lab of Nick Powell (Department of Metabolism, Digestion and Reproduction), they showed that the protein CXCL5 may be related to ulcerative colitis (a form of inflammatory bowel disease, IBD). They also found that some proteins have opposite effects on different autoimmune diseases. For example, the protein CD40 increases risk of rheumatoid arthritis, but appears protective against MS and inflammatory bowel disease.
Dr James Peters, from Imperial’s Department of Immunology and Inflammation, said: “This vast dataset builds our understanding of the predisposing molecular factors that lead to the development various autoimmune diseases. Although it can take years from identifying a drug target to a drug actually entering clinical practice, we hope that this data will ultimately provide the basis for new treatments.”
Read the paper in Nature Immunology.
Twisting light
 Chiral molecules are those that have two versions that are mirror images, like our right and left hands. These molecules have the same structure but different properties when they interact with other molecules, including those inside our bodies. This is important for example in drug molecules, where only the right- or left-handed version may have the desired effect.
Chiral molecules are those that have two versions that are mirror images, like our right and left hands. These molecules have the same structure but different properties when they interact with other molecules, including those inside our bodies. This is important for example in drug molecules, where only the right- or left-handed version may have the desired effect.
Detecting and quantifying the chirality of matter however has been difficult. Current methods using a form of light that produces a (right- or left-twisting) helix have the problem that each turn of the helix is much larger than the molecules. This creates important challenges for detecting molecular chirality.
Now, researchers at Imperial, with collaborators in Germany and Spain, have come up with a new way to use light to detect chirality. Instead of making light helix in space, they have devised a way to make it helix in time using lasers with moderate intensities. This form of light drives chiral electronic currents inside the molecules, causing one version of the molecule to emit bright light while its counterpart remains dark, vastly improving detection ability.
The team now plan to put the theory into practice, collaborating with other physicists at Imperial to use femtosecond laser facilities to image and manipulate chiral molecules.
Read the full paper in Science Advances.
 Want to be kept up to date on news at Imperial? Sign up for our free quick-read daily e-newsletter, Imperial Today.
Want to be kept up to date on news at Imperial? Sign up for our free quick-read daily e-newsletter, Imperial Today.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Hayley Dunning
Communications Division
Meesha Patel
Faculty of Medicine Centre