25 years of discoveries in cardiovascular research
We understand more about heart and circulatory conditions than ever before. A thriving cardiovascular research landscape has helped transform diagnosis, treatment, and care for millions of patients worldwide.
Yet there is still work to be done. Cardiovascular disease remains a major cause of mortality in the UK, responsible for a quarter of all deaths: equivalent to one death every three minutes.
Some important research questions remain unanswered: Is there a cure for heart failure? Can the heart repair itself after a heart attack? Can we correct faulty genes to permanently treat inherited heart conditions? Addressing these questions requires multidisciplinary approaches.
That’s why our research at Imperial’s Faculty of Medicine spans multiple departments in a way that truly fosters collaboration. Pivotal to this approach was the merger of several organisations that laid the foundations of the new Faculty of Medicine in 1997. This merger saw cardiovascular research brought together, from the National Heart and Lung Institute and the Royal Postgraduate Hospital Hammersmith, harnessing the collective strength of the Faculty’s research base.
Now the Faculty of Medicine is home to one of the UK’s largest grouping of cardiovascular scientists and clinicians dedicated to studying and treating cardiovascular conditions. This dedication has contributed to major discoveries and improvements for people living with cardiovascular conditions.
As the Faculty celebrates its Silver Jubilee, we reflect on how this collective strength has brought real benefits to society and changed our understanding of heart and circulatory health.
Tackling high blood pressure

Often described as a ‘silent killer’, high blood pressure – also known as hypertension – is one of the leading risk factors for cardiovascular disease.
Fortunately, high blood pressure is treatable and preventable. The benefits of lowering blood pressure have been well-recognised since the introduction of the first type of drugs in the 1950s.
The treatments have evolved considerably since then and a variety of options are now available. Several landmark Imperial studies have provided vital evidence for understanding what the best drugs are to treat high blood pressure and which patients can benefit from them.
Proving that statins reduce risk
Since the 1970s it has been clear that high blood pressure and high cholesterol are serious risk factors for heart disease and stroke. LDL (low-density lipoprotein) cholesterol, sometimes called ‘bad cholesterol’, can build-up in the walls of blood vessels and restrict blood flow.
The introduction of the cholesterol-lowering drugs known as statins in the 1980s sparked one of the longest-simmering medical debates on their benefits.
The evidence of statins benefiting those at very high risk of heart disease is clear-cut. However, questions were raised on whether statins reduce mortality for people with a lower risk of cardiovascular disease, and whether the benefits outweigh any side effects, most notably muscle pain.
In the late-1990s, Peter Sever – Professor of Clinical Pharmacology and Therapeutics at Imperial – led a pivotal trial that has influenced national and international guidelines for blood pressure and cholesterol-lowering medication ever since.
Imperial’s Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT) study came at a time when there was no clear indication of optimal treatment strategies to prevent cardiovascular disease in patients with high blood pressure.
“In simplistic terms, the trial set out to answer: are statins better than placebo, and are the newer blood pressure lowering drugs better than the most widely used combination therapy for high blood pressure at the time?” explains Professor Sever.
“The trial was a long time in the making as it took about ten years to raise the funds to run a trial of this scale with almost 20,000 patients over 32 sites in the UK, Ireland and Scandinavia,” he adds.
“The results of ASCOT were so important that they were incorporated into national and international guidelines and therefore changed clinical practice”
The results of the lipid-lowering arm of the trial revealed that both coronary events and strokes were significantly reduced by statins compared with placebo. As for the other arm of the trial, results showed that a combination of the newer blood pressure-lowering drugs was far more effective than the older traditional combination.
National guidelines in the UK now advocate the use of statins as part of the management strategy for the primary prevention for cardiovascular for those who are at high risk.
Decades on, the legacy of ASCOT is the huge amount of information that was generated in a trial of this scale.
“The data gathered from the trial formed the basis of over 150 publications – the vast majority led by Imperial clinicians and researchers – which is remarkable and a testament to the team involved,” said Professor Sever.
Below are other key studies that have shaped the treatment approaches for high blood pressure.
Late-1990s: Salt and blood pressure
One of the largest trials exploring the link between salt intake reduction and blood pressure. The INTERSALT study, led by Professor Paul Elliott and colleagues at the School of Public Health, showed that salt intake is well above recommended levels in most populations. The findings played an instrumental role in developing international guidelines and public policy on salt reduction strategies
2005: Treatment for pulmonary hypertension
In 2005, Professors Martin Wilkins and Lan Zhao first demonstrated the benefits of using sildenafil (more commonly known by its brand name, Viagra) to treat pulmonary hypertension (PAH) – high blood pressure in the blood vessels of the lungs. Today, sildenafil and related drugs are still the most prescribed drugs for the treatment of PAH.
2007: Treating blood pressure in the elderly
HYVET, led by Professor Christopher Bulpitt and Dr Ruth Peters, was the first large trial of blood pressure-lowering treatment in the very elderly. The trial showed for the first time the benefits of blood pressure-lowering drug treatment in people aged 80 years or older. The results from HYVET shaped high blood pressure guidelines in many nations.
2015 - 2021: Cholesterol-cutting treatment
Professor Kosh Ray designed a new of type treatment to lower cholesterol, offering patients a lasting reduction in LDL cholesterol and cutting the risk of heart attacks. The treatment called inclisiran works by ‘silencing’ genes.
In 2021, inclisiran was approved for administration in primary care settings as a twice-yearly injection to people with high cholesterol who have already had a previous cardiovascular event to reduce the chances of them having another.
2016: Highlighting the scale of the problem
In 2016, in what was the largest study of its kind, Imperial’s Global Environmental Health group analysed blood pressure in every nation between 1975 and 2015. This amounted to nearly 20 million people.
They found that the number of people with high blood pressure had almost doubled in 40 years to more than 1.1 billion worldwide. Most revealing was that the burden of the condition has shifted from the rich to the poor.
The research highlighted the regions where people are not getting the blood pressure treatment they need and provided policy-makers with vital evidence to improve access to treatments.
Preventing and treating the failing heart

In the mid-1990s, Imperial researchers embarked on a series of studies that changed the course of action on the use of beta-blockers for heart failure treatment.
Heart failure occurs when the heart muscle becomes weak and can’t pump blood as well as it should. Whilst there is currently no cure, it can be managed long-term with medications.
For 30 years, all international guidelines advised against the use of beta-blockers for heart failure as they were completely contraindicated up to the late 1990s.
Guided by evidence-based research, including that from Imperial, beta-blockers are now a worldwide mainstay of heart failure treatment and recommended in all international guidelines for chronic heart failure. So why the U-turn?
Professor Sian Harding has spent four decades working in cardiovascular research and played a key role in Imperial’s work to improve the understanding of beta blockers.
“The heart is supported by hormones such as adrenaline. Beta-blockers block the activity of these hormones and so decrease the activity of the heart. Up to the late-1990s, beta-blockers were contraindicated for heart failure because they initially worsened cardiac contraction and can in fact kill people who are in heart failure,” explains Professor Harding.
A turning point came in the early-1990s when it was demonstrated that beta-receptors actually become desensitised over time and subsequent studies showed that giving beta-blockers in carefully titrated doses to heart failure patients had miraculous long-term effects on their health and longevity.
Imperial researchers were pivotal in defining beta-adrenoceptor/beta-blocker mechanisms in failing human hearts and translating the benefits into clinical practice.
“I was mainly doing the scientific work and Professor Philip Poole-Wilson and Professor Andrew Coats at the Royal Brompton Hospital were running the trials, specifically COMET and the UK arm of the COPERNICUS trial,” recalls Professor Harding.
These studies helped establish beta-blockers as the mainstay in modern heart failure management.
The trials showed that although beta-blockers caused an initial depression of cardiac function in heart failure patients, continued use of the medication not only restored function but improved it. Most importantly, mortality was reduced by 30-40%.

Solving a genetic puzzle
Discovering new genes that cause cardiovascular conditions has allowed us to leap forward in protecting future generations.
One of the major areas where Imperial has had a considerable impact has been the genetic causes of inherited dilated cardiomyopathy (DCM). Progress in this area has been propelled by Imperial’s strong research base in genomic medicine.
Estimated to affect up to 1 in 250 people in the UK, DCM is a disease where the heart muscle becomes thinner and causes the heart to weaken, leading to heart failure. The condition can be inherited: around 30-50% of those affected have a family history of DCM.
At a time when it was deemed there was little role for genetic testing in DCM, researchers at Imperial helped to establish a link between inherited DCM and a faulty copy of the Titin (TTN) gene.
Titin is quite an unusual gene – it encodes for the largest protein in the human body.
In 2012, a global consortium involving Imperial researchers identified that DCM was commonly caused by a faulty copy of the TTN gene.
This pivotal discovery, backed by further studies led by Imperial's Professors James Ware and Stuart Cook, subsequently paved the way for a diagnostic test for changes in the TTN gene.
"Titin is so big that it is very common to have a genetic change – we did lots of work to figure out which variants were important, which was crucial for turning the discovery into a diagnostic test," recalls Professor James Ware.
Testing for changes in the TTN gene was first rolled out at the Clinical Genetics and Genomics laboratory based at Royal Brompton Hospital, and following a successful trial period, progressed to inclusion in all DCM genetic testing panels across the NHS.
As a result of the Imperial research, genetic testing is now routine practice for DCM. This represents a major improvement in the clinical management of patients with this condition.
Early data from the use of the TTN test was used to support the establishment of the Clinical Genetics and Genomics laboratory based at the Royal Brompton Hospital – one of only four in England that offers genetic testing for families and individuals at risk of inherited cardiac conditions.
Dr Deborah Morris-Rosendahl, a Consultant Clinical Scientist has been Head of the Clinical Genetics and Genomics Laboratory since its launch.
“We initially set up the laboratory in 2012 based on the findings of the Titin work but with a view to offering genetic testing for all inherited cardiac conditions. There was a clear clinical need for more genetic testing for inherited cardiac conditions. We now test for about 250 known genes that are associated with cardiac conditions, including all cardiomyopathies.
The implications of testing an individual patient are huge for the rest of the family.
“If we find the genetic cause of an inherited cardiac condition in a patient, we’ll then offer testing for all relevant family members. This can help refine a diagnosis for family members with similar symptoms or can predict the potential risk of them acquiring the same condition,” explains Dr Morris-Rosendahl.
Knowing an individual’s genetic profile can now help guide treatment choices, moving closer towards an era of personalised medicine.
“If you know that you're at risk for a cardiac condition, it means that you immediately get the correct clinical screening. There may even be a targeted therapy and/or preventive measures that can be taken. It’s a significant advantage to have that knowledge and also to be able to provide reassurance to those who are not at risk” adds Dr Morris-Rosendahl.
Improving diagnosis through technology

Driven by Imperial’s multidisciplinary approach, the fusion of skills and technologies has allowed us to develop innovative methods for disease detection – ultimately improving the patient experience.
Using machine learning to analyse heart scans
Recent advancements in artificial intelligence (AI) techniques are helping to push the boundaries of detection and risk identification in cardiovascular disease.
With cardiovascular disease steadily on the rise, what if we were able to predict and diagnose at-risk patients before the damage is done, enabling us to take preventative measures? This is the promise that may be offered through AI techniques, such as machine learning.
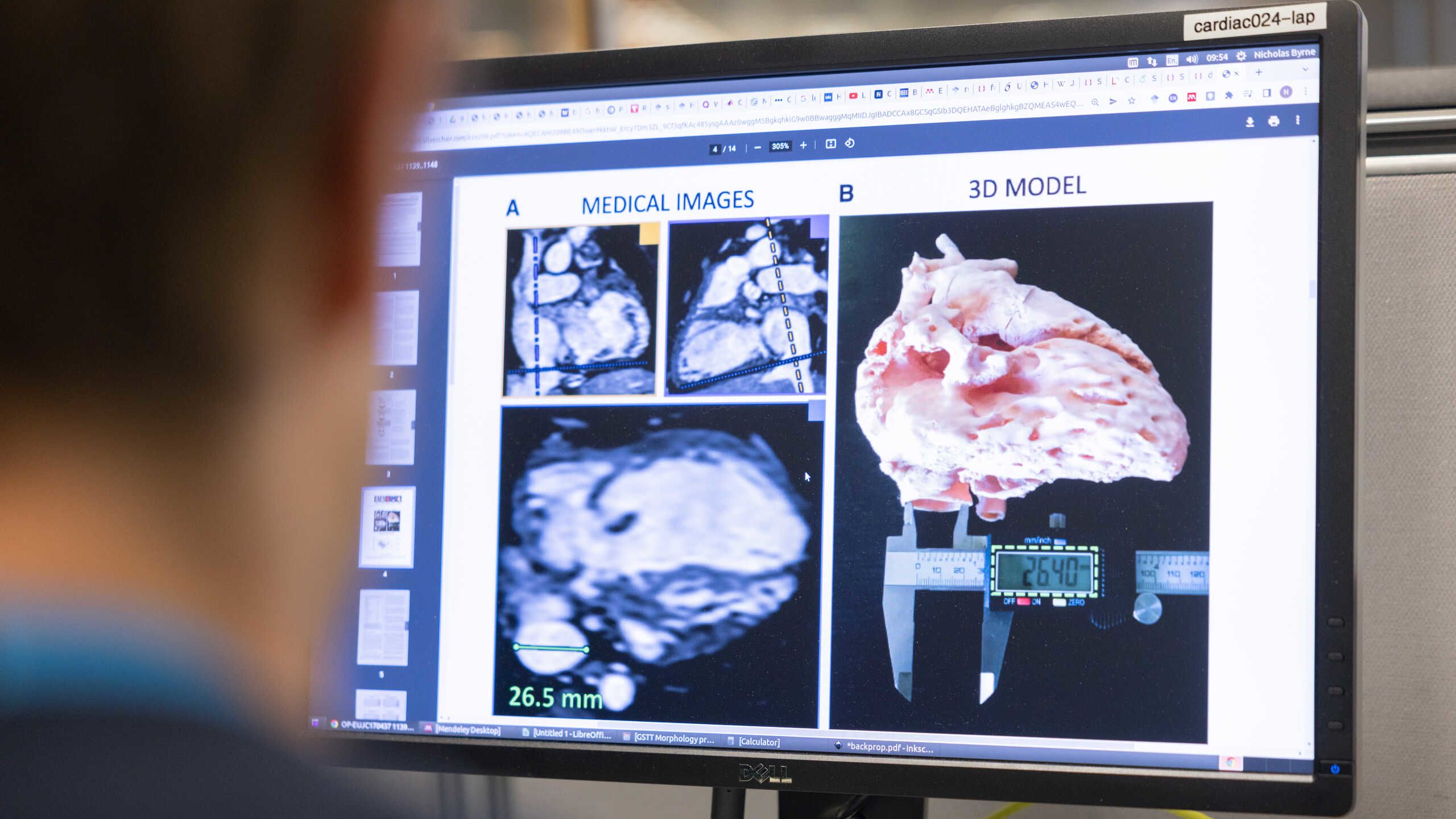
In 2017, Imperial clinicians working with colleagues at the Department of Computing developed software that showed that machine learning can predict death risk in people with serious heart disease faster and more accurately than current methods.
“This is the first time computers have interpreted heart scans to accurately predict how long patients will live. It could transform the way doctors treat heart patients
Using data from 250 heart failure patients, the software analysed moving MRI scans of each patient’s beating heart, replicating 30,000 points to create a ‘virtual 3D heart’. This automatically learned which features were the earliest predictors of heart failure and death.
To stent or not to stent?
Dr Justin Davies’ career as a clinical academic has seen him combine both his passions: interventional cardiology and computer programming.
From an initial idea born out of a cardiology-bioengineer collaboration, Dr Justin Davies, Professor Jamil Mayet and colleagues developed instantaneous wave-free ratio (iFR) – a system that is now used in cardiac catheter labs across the world.
The system assesses the severity of stenosis – narrowing of a patient’s arteries – to help clinicians decide if a patient requires a stent. If the stenoses aren’t severe, they can be left alone.
Compared to the main technique used at the time – fractional flow reserve (FFR) – iFR doesn’t require intravenous adenosine to relax the blood vessels. The other benefits of iFR are improved patient comfort and reduced cost.
First used in a select few patients in Hammersmith Hospital, the technology has since been licensed to Volcano-Philips and now used in over 5,000 cardiac catheter labs in more than 30 countries. As a result of the Imperial research, iFR is recommended in all the major cardiology guidelines.
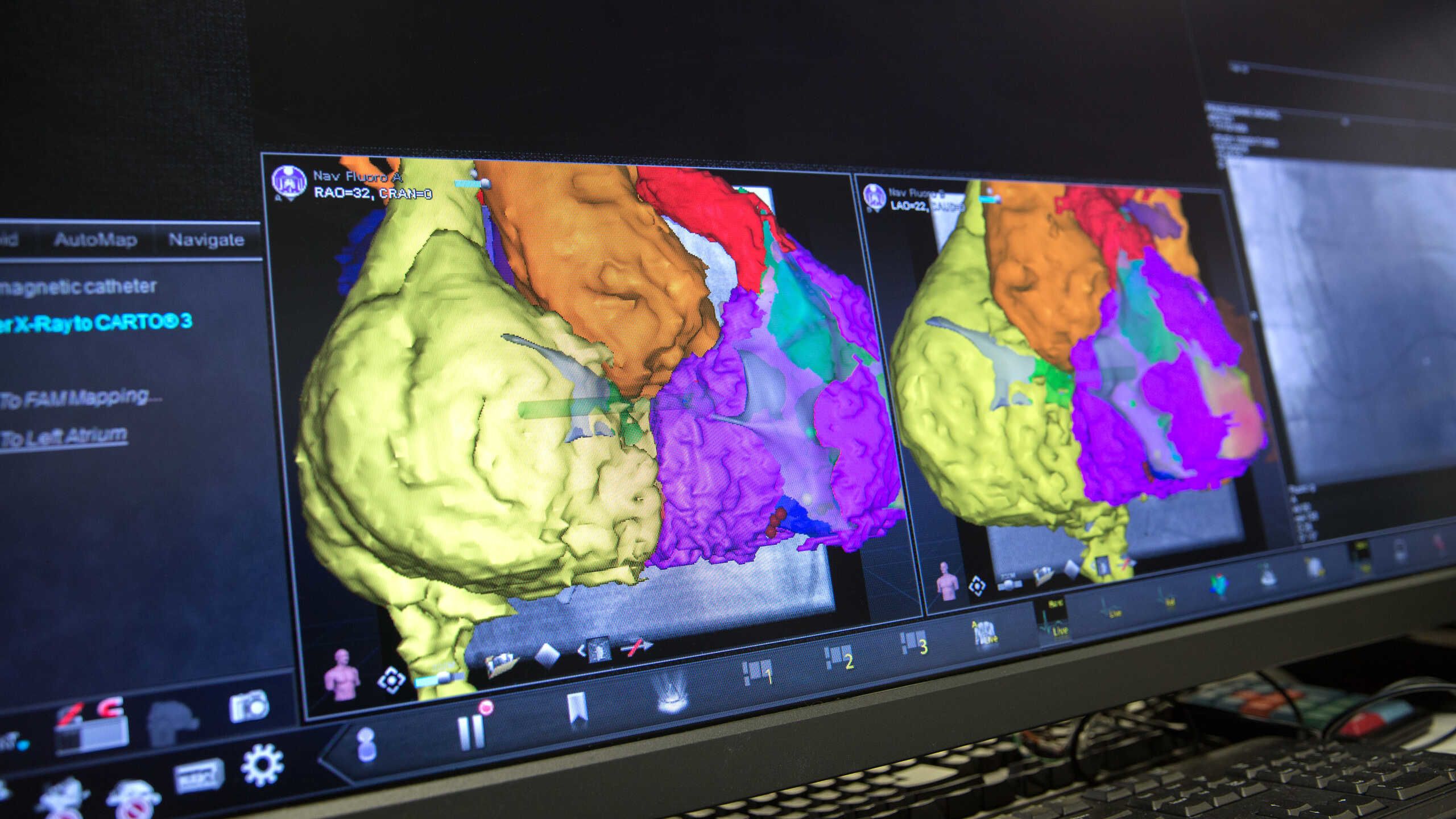
3D mapping of arrhythmias
In the late 2010s, a collaboration between Professor Prapa Kanagaratnam and colleagues from the Department of Bioengineering made a significant contribution to improving the treatment method of cardiac arrhythmias with the development of Ripple Mapping.
Arrhythmia is caused by a problem in the electrical conduction system that controls the heart. This can result in the heart beating too quickly (tachycardia), too slow (bradycardia) or irregularly (arrhythmias).
Successful treatment of cardiac arrhythmias through a method known as ablation relies on pinpointing the exact origin of the abnormal electrical activity.
The Ripple Mapping technique provides a visual 3D representation of the patient’s arrhythmia during an examination, making it easier for electrophysiologists to pinpoint the exact source of the abnormal electrical activity.
Ripple mapping increases the success rate and reduces the time needed for curative thermal ablation compared to conventional manual mapping.
Following this, Ripple Mapping was licensed and incorporated within their commercial anatomical electro-mapping system CARTO system, now installed in thousands of hospitals across the world.