Understanding how the brain keeps time
The Brancaccio Laboratory studies how the brain generates and maintains circadian time and why disruption of this process can compromise brain health. Nearly every aspect of physiology and behaviour follows daily rhythms, from sleep and metabolism to cognition and immune function. These rhythms are coordinated by the brain’s master circadian clock, located in the suprachiasmatic nucleus (SCN) of the hypothalamus.
Our research asks a fundamental question: how do neural circuits generate internal time?
Rather than acting as a simple molecular oscillator, the SCN is a complex network in which neurons, astrocytes, and molecular clocks interact to produce robust and synchronised rhythms. Understanding how these components cooperate to generate stable circadian signals—and how this organisation fails in disease—is a central goal of our work. (Movie 1)
Circadian imaging
By combining systems neuroscience, molecular biology, and advanced imaging approaches, we investigate circadian timing across multiple levels of biological organisation, from molecular clocks within single cells to activity patterns across neural circuits and behaviour.
Research Themes
Circadian circuits in the brain
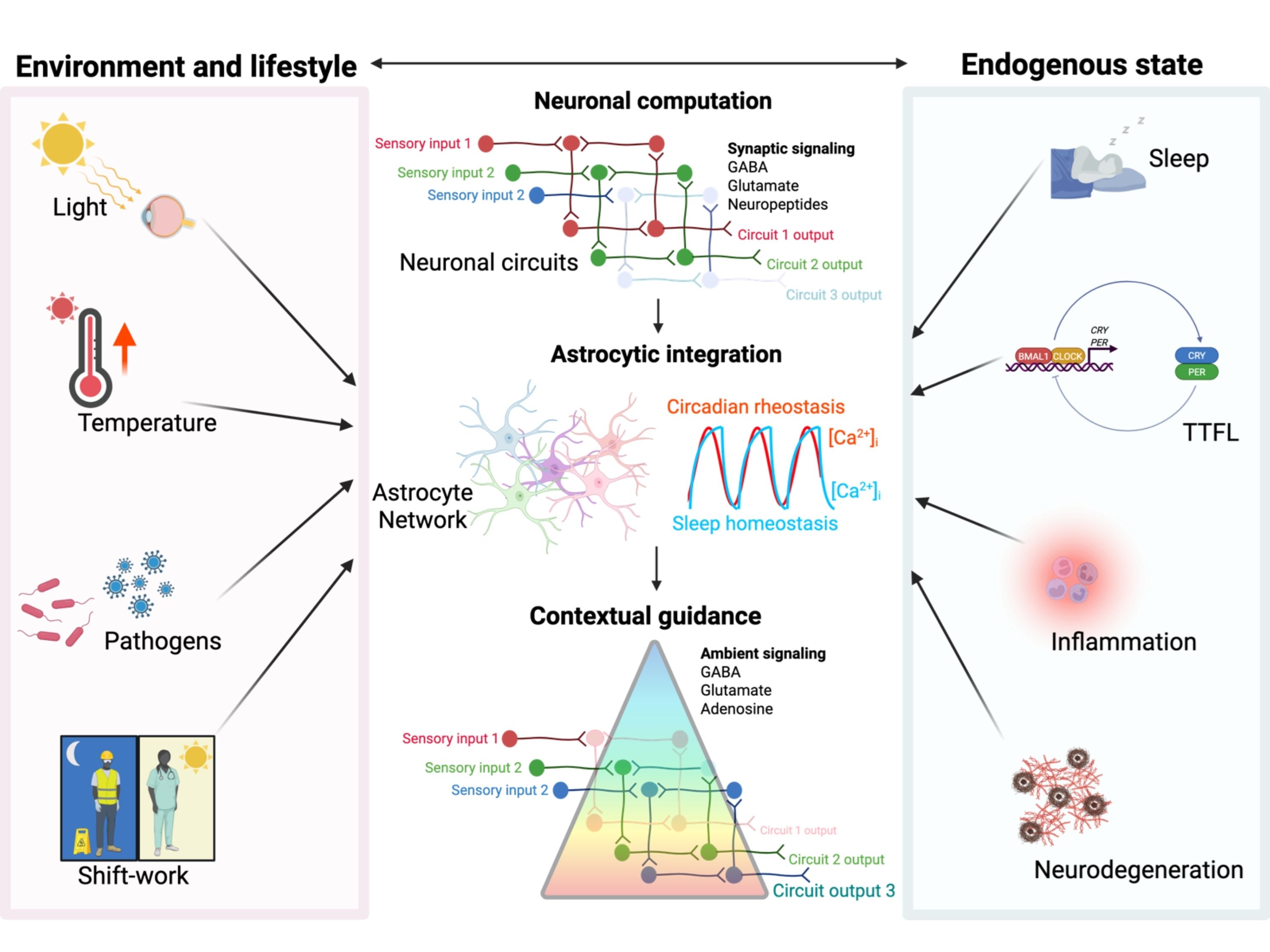
Circadian time in mammals is generated by the suprachiasmatic nucleus, a specialised neural circuit composed of thousands of interconnected neurons and glial cells. While each cell contains a molecular clock, coherent circadian rhythms emerge only through interactions across the circuit.
Our work investigates how neural and glial populations cooperate to generate robust rhythmic signals. We study how communication between cells, network organisation, and neurotransmitter signalling shape circadian timing. A particular focus of our research is understanding how astrocytes—long considered passive support cells—actively contribute to the generation and regulation of circadian rhythms.
By studying the SCN as a neural circuit, rather than simply a molecular clock, we aim to understand how the brain encodes and maintains temporal information.
Astrocytes and the architecture of circadian time
Astrocytes are increasingly recognised as key regulators of brain physiology. Our work has shown that astrocytes within the SCN contain their own circadian clocks and play an active role in controlling neuronal rhythms (Brancaccio et al. Neuron 2017; Brancaccio et al., Science 2019; Ness et al., EMBO J, 2025).
We investigate how astrocytes interact with neurons to regulate circadian timing, how metabolic and signalling pathways influence glial clock function, and how glial dysfunction may destabilise circadian circuits. By combining live imaging, genetic manipulation, and circuit analysis, we explore how neuron–glia communication contributes to the generation of daily rhythms in the brain.
Understanding these interactions provides insight into fundamental principles of neural circuit organisation and reveals new mechanisms through which circadian disruption may affect brain health.
 Brancaccio Bioessays 2025
Brancaccio Bioessays 2025
Circadian disruption and neurodegeneration
Sleep and circadian disturbances are among the earliest symptoms of many neurodegenerative disorders, including Alzheimer’s disease and frontotemporal dementia. These changes often emerge years before measurable neurodegeneration or cognitive decline.
Our laboratory investigates whether circadian dysfunction is simply a symptom of disease or whether it actively contributes to disease progression. We examine how pathological proteins such as amyloid and tau interact with circadian circuits, how disruption of SCN function affects memory-related brain networks, and whether restoring circadian regulation can stabilise brain homeostasis.
This research bridges chronobiology and neurodegeneration and aims to identify mechanisms through which circadian disruption increases vulnerability of neural circuits.
Experimental approaches

To understand circadian circuits, we use a multipronged approach. Our laboratory develops and uses techniques that allow us to observe and manipulate neural networks across time and biological scales.
These include viral vector engineering and somatic genetic manipulation, long-term live imaging of circadian activity in brain tissue, fibre photometry recordings in freely behaving animals, sleep and behavioural monitoring in naturalistic conditions, and molecular profiling approaches such as transcriptomics and proteomics. Finally, we use preclinical human model for the translation of findings, including patient-derived hIPSC-derived neural cells.
By integrating these approaches, we can link molecular mechanisms to circuit dynamics and behaviour, providing a systems-level understanding of circadian biology.
In their Words
Marieke, Swiss National Science Foundation Fellow, Postdoc
“When we sleep, the temperature in our brain decreases, stimulating expression of cold-induced proteins. We want to test if these proteins contribute to the restorative effects of sleep on neuronal function and cognition and help us getting ready for the new day. Can we harvest the benefits of sleep and protect from cognitive decline observed in neurodegenerative diseases by recruiting these ancient allies?”
Anne, UK DRI Postdoc
"My aim is to decipher the synaptic and extra-synaptic mechanisms by which astrocytes regulate daily plastic changes of neuronal function in the adult and ageing brain. Astrocytes may hold the key to revert the depression of circadian function in the brain observed during ageing and dementia processes - a versatile target for future therapeutic interventions."
In their Words
Renaud, UK DRI Postdoc
“One of the most important function of our circadian clocks is to control inflammatory processes in our body, day on day. Yet, we know very little about how the principal brain resident immune cells, microglia, are dynamically regulated during the day. As they are found to play a Dr Jekyll and Mr Hyde in Alzheimer’s disease, can we use the molecular clocks to make them show us their best face?”
Natalie, President’s PhD Scholar
"I want to explore how astrocytes and microglia interact to affect synapse function in neuronal circuits driving circadian behaviour. As glia modulate the daily plasticity of synapses, can we harness glial clocks to counteract the circadian network dysfunction and disrupted sleep-wake cycles observed in neurodegenerative diseases?"
General enquiries
For any enquiries about our lab, please contact:
Dr Marco Brancaccio
m.brancaccio@imperial.ac.uk
+44 (0)20 7594 7907
