Quality Assurance and Regulatory Affairs (QARA) support is a core component of the MedTech Accelerator. Medical technologies are developed within a highly regulated environment, and early regulatory planning is essential to avoid delays, redesign, or unsuccessful submissions later in development.
Our QARA function helps research teams navigate UK, EU, and international regulatory frameworks while embedding quality management practices from the earliest stages of technology development.
Regulatory pathway guidance
We support teams in understanding and planning their regulatory strategy, including:
-
determining medical device classification
-
understanding MHRA, FDA, and CE/UKCA requirements
-
identifying relevant standards (e.g. ISO 13485, ISO 14971, IEC 62366)
-
planning design controls and verification strategies
-
aligning regulatory milestones with technology development stages
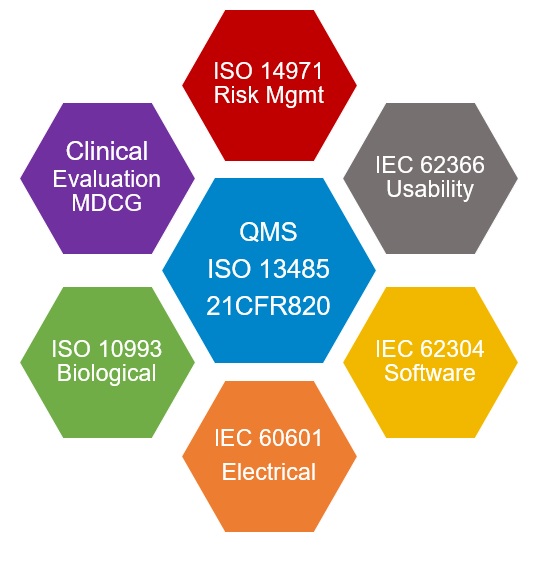
Quality management systems
Teams have access to an ISO 13485-aligned Quality Management System (QMS), enabling structured documentation, risk management, design controls, and preparation of technical documentation required for regulatory submissions.
Regulatory readiness
Using the MedTechONE Regulatory Readiness Level (RRL) framework, we assess a project’s regulatory maturity and identify the steps required to progress towards clinical investigation, regulatory approval, or commercial translation.
Practical regulatory support
Our QARA specialists provide hands-on guidance in areas such as:
-
defining intended purpose and indications for use
-
mapping safety and performance requirements
-
developing technical documentation
-
preparing for MHRA, FDA, or Notified Body engagement
-
planning clinical evaluation strategies
Enabling safe and compliant innovation
Working closely with translational project managers, IPC, and clinical partners across Imperial, the QARA function ensures that regulatory and quality considerations are integrated into project planning from the outset—helping technologies progress efficiently towards clinical use, licensing, or spin-out.
