Scientists uncover drug targets for arthritis and inflammatory kidney disease
by Sam Wong
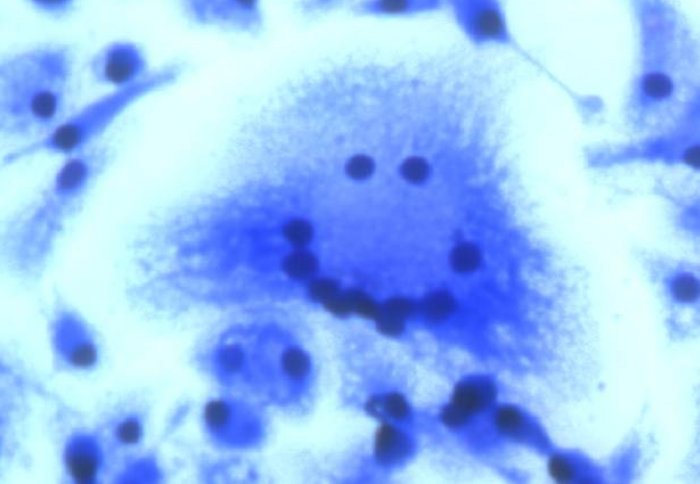
The drug target Kcnn4 controls the formation of giant cells, like this one whose nuclei form a smiling face.
Scientists at Imperial College London have discovered new drug targets to treat inflammatory disease by studying the "social networks" of genes.
Using a new “systems genetics” approach, the scientists identified a large network of genes that work together in cells to control processes involved in inflammatory disease. By targeting one specific molecule involved in this network, Kcnn4, the researchers showed that they can significantly reduce the symptoms of arthritis and kidney inflammation in animals.
Kcnn4 has previously been shown to be involved in immune system responses. Drugs to block the Kcnn4 molecule are currently in phase III clinical trials to target diseases in which the immune system goes wrong, such as inflammatory bowel disease and asthma. This study identifies two new potential uses for these drugs in preventing debilitating inflammatory diseases.
Kcnn4 is a molecule that is found on the surface of infection-fighting cells called macrophages. Macrophages can fuse together into a single big cell, called a giant cell, which contributes to various inflammatory diseases. The research, published in scientific journal Cell Reports, has shown for the first time that Kcnn4 works by controlling the formation of giant cells. It also shows that if the macrophages are treated with drugs to block the action of the Kcnn4 molecule, the cells do not fuse together and therefore inflammatory symptoms are reduced.
This work is fundamental to understand how genetic differences between individuals can affect the ability of their macrophages to fuse.
– Dr Jacques Behmoaras
Department of Medicine
In bones, these giant macrophage cells eat away at bone tissue and reabsorb the nutrients into the body. In normal circumstances, bone is constantly being built up again so it remains unharmed by this process. However, in arthritis patients with inflamed joints giant cells can lead to bones becoming less dense and more fragile.
In collaboration with researchers at Yale University, the authors showed that treating mice with drugs that target Kcnn4 to stop the giant cells forming alleviates the symptoms of arthritis by preventing bone erosion and inflammation.
Giant cells also contribute to an inflammatory kidney disease called glomerulonephritis, which is a common cause of kidney failure. Administering drugs that stop the action of the Kcnn4 molecule relieved this disease in rats by preventing the formation of giant cells in the kidney.
The researchers confirmed that the same network of genes that controls the formation of giant cells in rodents is also present in humans. Further studies would be required to prove that targeting Kcnn4 with drugs could ease inflammatory disease in humans.
Dr Enrico Petretto, from the Medical Research Council Clinical Sciences Centre at Imperial College London, said: “This study shows how our new ‘systems genetics’ approach has great potential to discover new genes that can be targeted to treat disease. Similar to what happens in social networks, Kcnn4 interacts with other genes inside a cell, forming a complex ‘gene network’. We discovered another 183 genes which could also prove to be promising drug targets for inflammatory disease. This study represents an important step towards understanding of the complex interactions in inflammatory disease.”
Dr Jacques Behmoaras, from the Department of Medicine at Imperial College London, said: “We have shown that Kcnn4 is a promising drug target for kidney inflammation. The fusion of macrophages giving rise to giant cells is still a poorly understood phenomenon. This work is fundamental to understand how genetic differences between individuals can affect the ability of their macrophages to fuse. Such information could be crucial to understand the genetic basis of inflammatory diseases.”
Professor Dorian Haskard, head of the Division of Immunology and Inflammation in the Department of Medicine, said: “Linking macrophage function to inflammatory disease by using genetics is a very promising approach. Understanding the genetic determinants of macrophage activation will facilitate the design of new treatments for important inflammatory diseases. The study by the Behmoaras group provides a nice example for identifying new pathways that could be potential new drug targets.”
Based on a news release from the Medical Research Council.
Reference: Kang et al (2014) 'Kcnn4 is a Regulator of Macrophage Multinucleation in Bone Homeostasis and Inflammatory Disease.' Cell Reports. DOI: http://dx.doi.org/10.1016/j.celrep.2014.07.032
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Sam Wong
School of Professional Development