Potential way to target cancer cells found using molecule which binds DNA knots
by Ben Lewis
A new method to bind to knots of DNA only in certain conditions could open doors to targeting cancer cells more specifically.
Whilst most DNA found in cells is arranged in an intertwined double-helix structure, it is possible for DNA to be found in other forms. One structure which has gained increasing interest in recent years is the G-quadruplex, where DNA forms small knots at specific points. It is believed these knots could play an important role in how our cells express our genetic code.
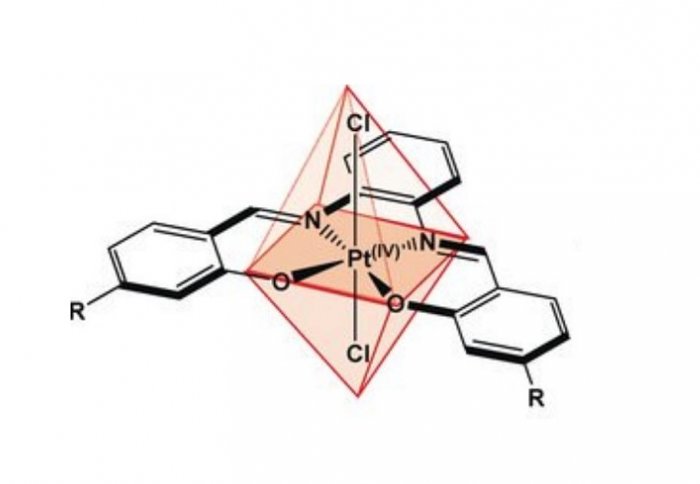
New research carried out at Imperial College led by Professor Ramon Vilar has produced a molecule which binds to these DNA knots only where there is very little oxygen - a condition which is most commonly found inside tumour cells, where quick growth means that many cells are far away from the blood vessels which supply their oxygen.
This new molecule, based around an atom of platinum, is at first in a form which cannot bind to DNA, but which is converted into a structure which can in environments with very little oxygen, such as in tumours. By binding to the DNA knots, they become more stable and this could potentially get in the way of the cell reading the genetic information contained there.
Targeting these knots has been considered as a way of killing cancer cells in particular for a long time because the knots are known to occur around parts of the genetic code which are important for the tumour cells to continue growing and dividing. By stabilising the knots, the tumour may not be able to continue growing and the cancer could be successfully treated, whilst making sure only to target knots in cancer cells would reduce the possibility of affecting other, healthy cells.
“A Redox-Activated G-Quadruplex DNA Binder Based on a Platinum(IV)–Salphen Complex” (DOI: 10.1002/anie.201709968) was published in Angewandte Chemie International Edition on 2 January 2018.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Ben Lewis
Department of Surgery & Cancer