Scientists discover new approach to fighting antibiotic resistance
A new approach to fighting antibiotic resistance could help to prevent diseases by making bacteria vulnerable to treatment again.
Researchers, including experts from Imperial College London, have found a way to impair antibiotic-resistant bacteria that cause human disease, such as E. coli, K. pneumoniae and P. aeruginosa, by inhibiting a protein that drives the formation of resistance capabilities within the bacteria.
Dr Despoina Mavridou, currently an assistant professor in Molecular Biosciences at the University of Texas at Austin, who led the research team, said the approach represented a “completely new way of thinking about targeting resistance,” which is a major health concern for scientists.
A study published in The Lancet in January found that antimicrobial resistance was the direct cause of at least 1.27 million deaths globally in 2019 and there are concerns about bacteria becoming increasingly resistant to existing antibiotics, with researchers struggling to identify new alternative drugs.
"Since the discovery of new antibiotics is challenging, it is crucial to develop ways to prolong the lifespan of existing antimicrobials." Dr Chris Furniss Department of Life Sciences, Imperial College London
Antibiotic resistant bacteria have a host of different proteins in their arsenals that neutralise antibiotics. To function properly, these resistance proteins have to be folded into the right shapes. Dr Mavridou’s research team found that a protein in bacteria called DsbA helps fold resistance proteins into the right shapes to neutralise antibiotics.
For their proof-of-concept study, published in the journal eLife, the researchers inhibited DsbA, using chemicals that cannot be used directly in human patients, to prevent the formation of resistance proteins.
The team is now planning to work on developing inhibitors that can be safely used in humans while also achieving the same protective effect.
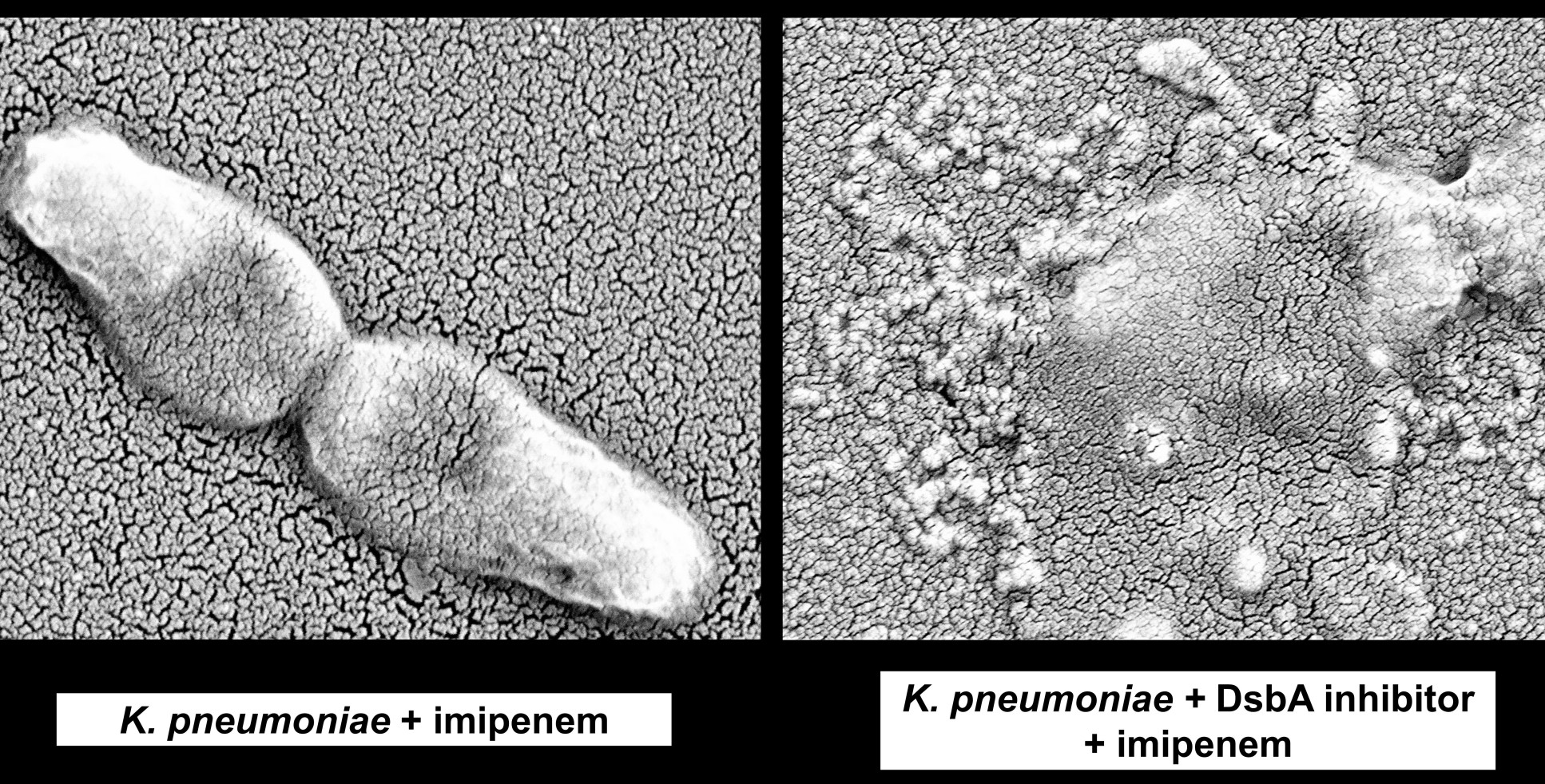
Dr Chris Furniss, one of the lead authors for the study from the Department of Life Sciences at Imperial, said: “Since the discovery of new antibiotics is challenging, it is crucial to develop ways to prolong the lifespan of existing antimicrobials.
“Our findings show that by targeting disulfide bond formation and protein folding, it is possible to reverse antibiotic resistance across several major pathogens and resistance mechanisms.
“This means that the development of clinically useful DsbA inhibitors in the future could offer a new way to treat resistant infections using currently available antibiotics.”
The researchers hope to ultimately combine a DsbA inhibitor with existing antibiotics to restore the drugs’ ability to kill bacteria.
International collaboration
Scientists had previously known that DsbA was involved in a range of functions in pathogens, such as building toxins to attack host cells and assisting with the assembly of needle-like systems that can deliver these toxins into human cells and cause disease.
Dr Mavridou began investigating the possibility that DsbA played a key role in the folding of proteins that help bacteria resist antibiotics while she was an MRC Career Development Fellow at Imperial College London, and before moving to the University of Texas at Austin (UT Austin) faculty in 2020.
Nikol Kaderábková, previously a graduate student at Imperial and currently a postdoctoral researcher at UT Austin, and the second lead author of the study, said: “We reasoned that if DsbA is required for the folding of resistance proteins, preventing it from working would indirectly inhibit their function.”
Other researchers involved in the study were based at Universidad de Sevilla in Spain, Brunel University London, the University of Birmingham, Paris-Sud University in France, and Université de Neuchâtel in Switzerland.
This research was supported in part by the Medical Research Council in the UK and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health in the US.
-
‘Breaking antimicrobial resistance by disrupting extracytoplasmic protein folding’ by R Christopher D Furniss et al. is published in eLife.
Article supporters
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Conrad Duncan
Communications Division