
Health
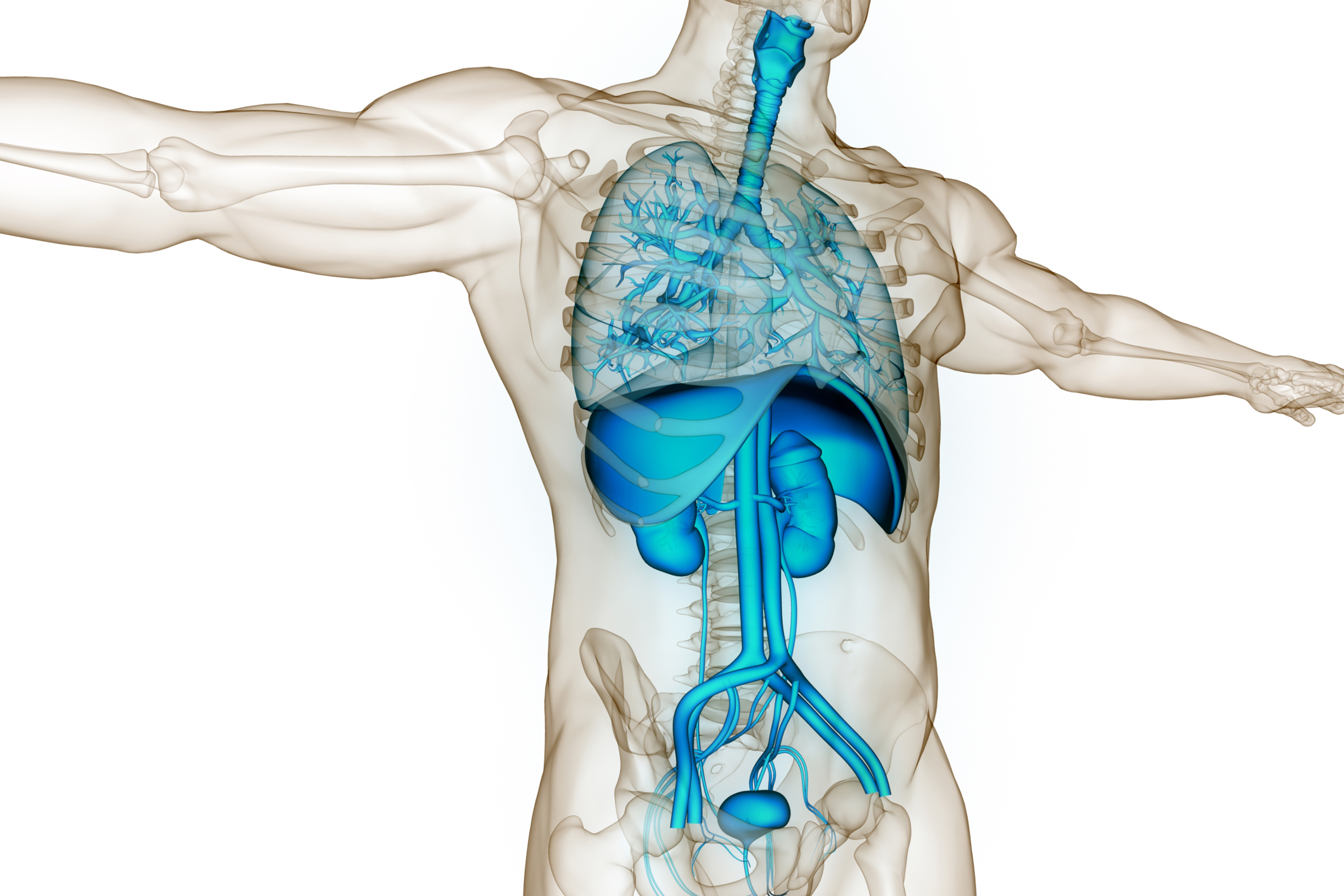
A new research hub will create open-source computer models of organs to help accelerate the development of new medicines.
The Modelling-Informed Medicine Centre (MiMeC), founded by the biopharma company GSK and world-leading universities Imperial College London and the University of Oxford, will provide a new UK hub for research in the emerging modelling-informed medicine field.
We have seen maths used for modelling aeroplanes and cars – and increasingly there is a realisation that you can perform virtual experiments in models of humans at great speed. Professor Steven Niederer Imperial
The centre will create computer models or ‘digital twins’ of organs and diseases to better understand how diseases of the lungs, liver and kidneys progress, to discover and develop drugs more quickly, and to target medicines more precisely.
It is backed by £11 million funding from GSK and multidisciplinary expertise spanning mathematics, data science, and experimentation from the founding partners.
The partners aim to support the life sciences community by bringing together fragmented research in the field and training a new generation of research and development specialists who understand best practice in this emerging area of biomedical research. It will share its models on an open-source basis and build collaborations with further partners.
GSK plans to use the research to incorporate models of organs into its drug development pipeline within five years, aided by industrial placements it will provide to researchers from the centre.
The programme is led by Professor Steven Niederer at Imperial, Professors Helen Byrne and Philip Maini at the University of Oxford, and Dr Anna Sher at GSK.
Professor Steven Niederer, from Imperial's National Heart and Lung Institute, said: “We have seen maths used for modelling aeroplanes and cars – and increasingly there is a realisation that this has benefits in biology, where you can perform virtual experiments in models of humans at great speed and a fraction of the usual cost.”
At Imperial, Professor Niederer and team will build patient-specific models of organs using artificial intelligence and biological datasets.
They will mathematically represent millions of cells in organs such as the lungs, and the mechanistic (or cause-and-effect) relationships they hold to one another, by modelling a proportion of cells found in the real organ.
Using the models, researchers could perform a simple in vitro experiment into the effect of a drug on a single lung cell and then use the model to simulate how this would translate into larger effects such as changes in the behaviour of the airways.
In contrast with computational approaches that merely find statistical regularities in biological data, these mechanistic models represent cause and effect, making them potentially more explainable and robust.
Eventually, the approach could allow clinicians to use digital twins of specific patients to tailor their treatments in real time, an approach that Professor Niederer’s group is already testing with cardiac patients.
At the University of Oxford, experts will develop and apply mechanistic models to advance understanding of disease processes and inform the design of more effective treatments.
The teams will build models grounded in physics, physiology and pharmacology to reveal disease mechanisms. These will include multi-scale models that integrate molecular, cellular and organ-level processes with whole-body physiology.
They will use digital twins and virtual patients to simulate treatment responses, optimise dosing strategies, and design in-silico clinical trials. They will also contribute open-source tools, standards for reproducibility, and case studies that showcase the impact of model-informed drug development.
Professor Jon Chapman, head of the Mathematical Institute at the University of Oxford, said: “This exciting new partnership recognises the pioneering role that the Wolfson Centre for Mathematical Biology has played – and continues to play – in applying mathematics to understand diseases and their response to treatment.”
MiMeC will focus on the adoption of the mathematical modelling-first mindset in the development of new therapies.
Dr Anna Sher, MiMeC Co-Director and Quantitative Systems Pharmacology lead in the Respiratory, Immunology and Inflammation Research Unit at GSK, said: “By cycling between computer modelling, learning from the results, making predictions and then testing them, we can make faster, better decisions in developing new medicines. The tools and models developed through MiMeC strengthen GSK’s ability to generate virtual patients and digital twins to run computer‑based (in silico) clinical trials, analyse different data types, and test scientific ideas more efficiently.”
Bringing the mechanistic modelling mindset to the forefront of quantitative medicine has the potential to help supercharge the UK life science industry.
Alex Elkins, Co-Director of Enterprise (Partnerships) at Imperial, said: “Alongside other recent significant investments by the pharmaceutical industry into Imperial research, the significant funding and close collaboration by our partners at GSK on this programme is a great vote of confidence in the quality of the science and our translation pathways at leading UK universities, which are helping create exciting advances in medicine.”
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Administration/Non-faculty departments


Health
.jpg)
Health

Cross-faculty

Health
Engineering
Imperial Global
Engineering
Discover more Imperial News
Search all articlesDiscover more Imperial News
Search all articles