
Health
by Dara O'Hare
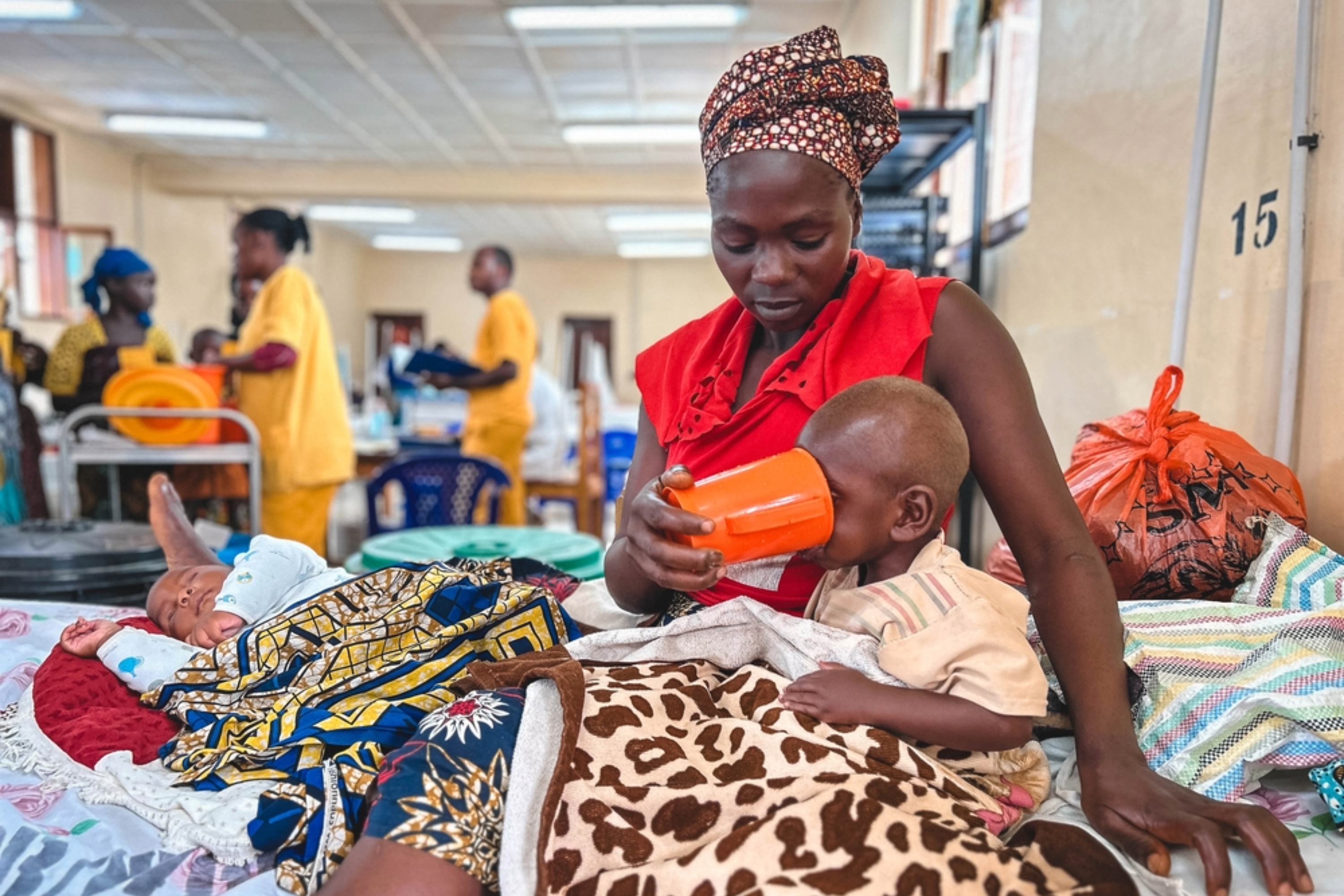
A major new clinical trial, conducted on over 400 children in four African countries, challenges longstanding international guidelines on how to treat dehydrated children with severe acute malnutrition caused by diarrhoea.
This has major clinical implications, given that diarrhoea is a critical health issue in sub-Saharan Africa and one of the leading causes of death among children under five. Children with severe acute malnutrition are at substantially higher risk of death when dehydrated.
According to UNICEF, in 2020, there were over 1 billion cases and more than half a million deaths from diarrhoea in the sub-Saharan region.
The findings of this second study from the Gastroenteritis in Severe Malnutrition (GASTROSAM) trial, published today in The Lancet Child and Adolescent Health, indicate that treating affected children with a standard formula of oral rehydration salts (WHO-ORS) – a diluted mix of salt and electrolytes – is just as safe and effective as the current recommended treatment – a low-sodium rehydration solution (ReSoMal).
If adopted, the findings could simplify global treatment protocols and increase access to life-saving rehydration therapy in low-income countries because WHO-ORS is widely available, inexpensive, and already used globally to treat children without malnutrition.
ReSoMal, conversely, is often unavailable in African hospitals and the guidelines recommend its use to treat children with severe malnutrition should be done under strict medical supervision.
For more than 30 years, international guidance has advised against using WHO-ORS because of concerns about sodium overload and potential heart failure. Instead, clinicians have been advised to use ReSoMal. However, these recommendations were not supported by strong trial evidence, particularly in Africa.
GASTROSAM was a phase 2, randomised controlled trial conducted across six hospitals in Kenya, Niger, Nigeria and Uganda. A total of 415 children aged 6 months to 12 years with severe acute malnutrition and moderate or severe dehydration due to diarrhoea were enrolled between 2019 and 2024.
They were randomly assigned to receive either ReSoMal (206 children) or WHO-ORS (209 children). They were followed for 28 days, with 97 per cent of participants remaining in the study all the way through.
The trial, led by Professor Kathryn Maitland, Director of the Centre of African Research and Engagement, found:
These findings suggest that WHO-ORS can be used safely in severely malnourished children. The authors recommend that protocols be simplified in order to:
Our findings show that standard WHO-ORS is safe in severely malnourished children and does not cause fluid or sodium overload. These results support simplifying treatment guidelines and could help close a major therapeutic gap in African hospitals where ReSoMal is frequently unavailable. Professor Kath Maitland Director, Centre of African Research and Engagement
Dr Johanna Thomson, Leader of the Médecins Sans Frontières International Paediatric Team, and one of the authors, said:
“This study challenges the long-standing dogma that standard oral rehydration therapy is unsafe for children with severe acute malnutrition. Simplifying rehydration protocols could make treatment safer, more consistent, and more accessible for children in resource-limited settings.”
Dr San-Maurice Quattara, an epidemiologist with Médecins Sans Frontières and another author, added:
“If the recommendation of this study were to be adopted, it would significantly transform access to safe and effective rehydration for severely malnourished children suffering from dehydration.”
Dr Elizabeth George, another author and the trial statistician, based at University College London, added:
"Despite the challenging conditions, the trial was carried out extremely carefully, to a very high standard and nearly all participants remained in the study for the full 28 days. This is a reflection of how committed the local teams were and how hard they worked to support everyone involved."
This trial builds on earlier GASTROSAM findings on intravenous rehydration published in the New England Journal of Medicine in 2025.
The study was funded by the Joint Global Health Trials Scheme (UK Medical Research Council, UK Department for International Development, and Wellcome) and Médecins Sans Frontières.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Faculty of Medicine


Health

Engineering

Health

Imperial Global
Health
Health
Engineering
Discover more Imperial News
Search all articlesDiscover more Imperial News
Search all articles