Hidden drug reservoirs in cancer cells could explain treatment resistance
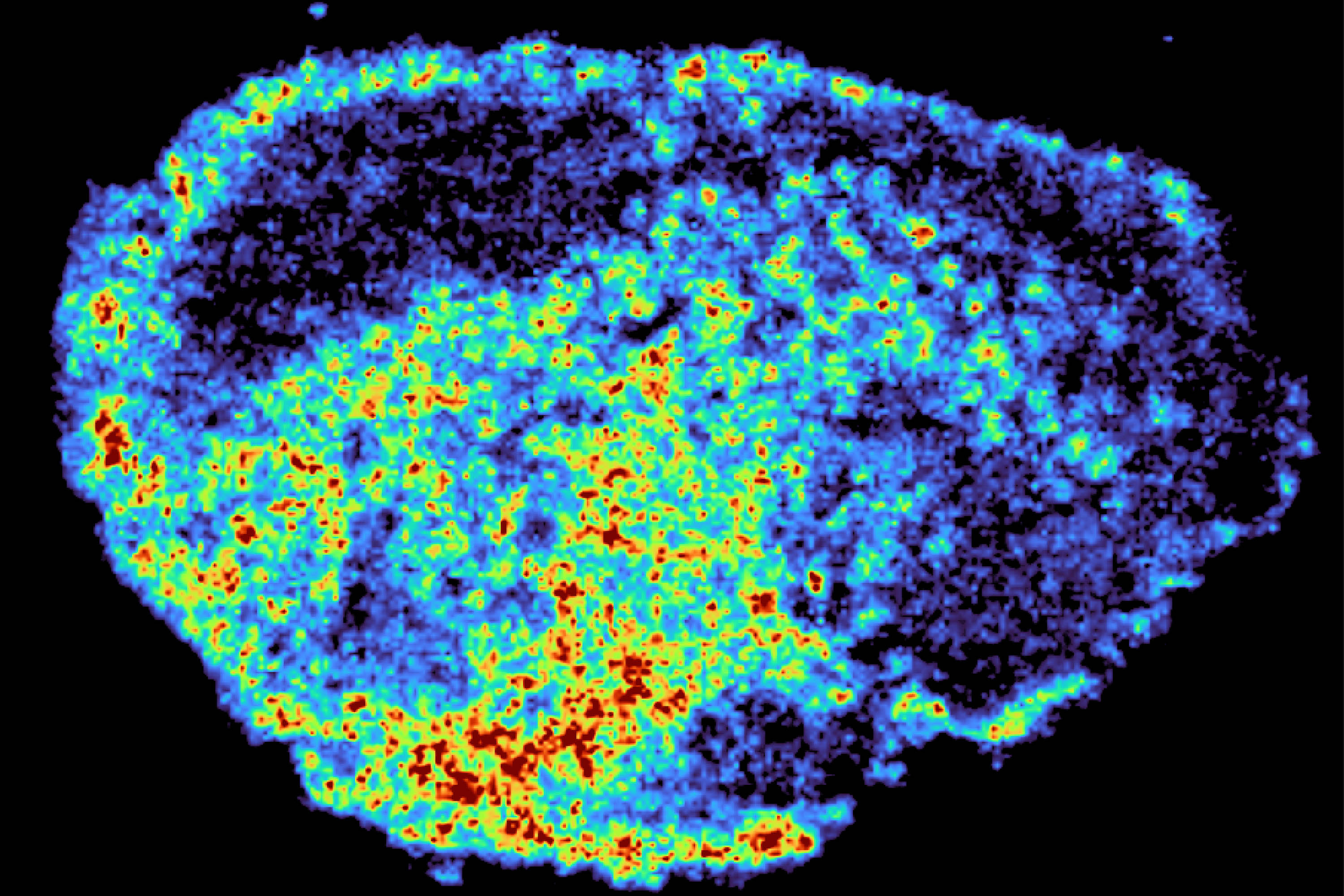
Cancer drugs can accumulate in hidden “storage hubs” inside tumour cells, creating uneven drug exposure across tumours, according to new research from the MRC Laboratory of Medical Sciences (LMS) and Imperial College London.
A novel aspect of this study was the use of mass spectrometry imaging to directly measure and visualise drug uptake in patient tumour tissue. Dr Zoe Hall, Senior Author Associate Professor at the Department of Metabolism, Digestion and Reproduction
This means some cancer cells receive much higher drug doses than others, helping explain why treatments work well for some patients but not others, and opens avenues towards more personalised approaches to cancer therapy in the future.
One of oncology’s biggest challenges is that the same treatment can work well for some patients but fail completely in others. A study published in Nature Communications, from a multi-disciplinary team led by Dr Louise Fets at the LMS and including Dr Zoe Hall from Imperial’s Department of Metabolism, Digestion and Reproduction, has mapped the distribution of a type of targeted treatments, known as PARP inhibitors, using advanced imaging techniques and patient ovarian tumour samples.
The research reveals that build up of these drugs in lysosomes – small compartments inside cells that normally act as “recycling centres” – can trap and release certain drugs over time, shaping how well treatments work.
Mapping drug delivery
Cancer treatment options have increased exponentially in recent years, significantly improving patient prognosis. For ovarian cancer patients, this revolution has come in the form of a group of drugs called PARP inhibitors. However, some patients do not respond to treatment, while others develop resistance over time. For a drug to be effective, it needs to build up in cancer cells at sufficient levels to trigger cell death. Despite its importance, drug distribution within tumours, and the mechanisms that regulate this, are poorly understood.
This study shows that the problem isn’t just whether a drug reaches a tumour, but how it spreads within the tumour and inside its cells. The researchers used thin slices of ovarian tumours taken from patients and kept alive in the lab. These tumour “explants” were treated with PARP inhibitors, allowing the team to directly observe how drugs spread through real human tumour tissue.
This was possible thanks to a collaboration with colleagues from Imperial's Department of Surgery and Cancer. The tumour samples were obtained from patients undergoing cytoreductive surgery at Hammersmith Hospital, performed by Professor Christina Fotopoulou, and were maintained using a patient-derived explant (PDE) culture system established by Dr Paula Cunnea.
Using mass spectrometry imaging the team created high-resolution maps showing exactly where drug molecules accumulated. They combined this with spatial transcriptomics, which allowed them to compare gene activity in regions with high and low drug levels, all within the same tissue sample. The results showed drug levels varied dramatically across different regions of the same tumour and between patients, even when the same drug dose was used.
Dr Zoe Hall, senior author and Associate Professor at Imperial’s Department of Metabolism, Digestion and Reproduction, said: “A novel aspect of this study was the use of mass spectrometry imaging to directly measure and visualise drug uptake in patient tumour tissue. Through the spatial mapping of drug molecules, we could pinpoint regions of high and low drug and compare gene expression, from the same tissue slice, using spatial transcriptomics.”
Lysosomes: the cell’s hidden drug storehouses
The team discovered that the uneven drug distribution was driven by lysosomes. Some PARP inhibitors were being pulled into lysosomes and stored there. Instead of spreading evenly through the cell, the drugs became trapped in these compartments, creating internal drug reservoirs. These lysosomal reservoirs act like slow-release stores, holding the drug and releasing it over time, which increases exposure in some cells, while leaving others relatively unexposed. To further complicate matters, not all PARP inhibitors behave the same way. The study found that this lysosomal storage mechanism applies to some drugs, such as rucaparib and niraparib, but not others like olaparib.
Dr Carmen Ramirez Moncayo, first author and Postdoctoral Researcher at the LMS, said: “We were surprised to see large variability in drug accumulation at the single-cell level. This variability was driven by the build-up of a drug in lysosomes, which are acting as reservoirs, increasing the exposure of cancer cells to drugs, by storing and releasing the drug when needed.”
The future of cancer treatment
PARP inhibitors are already widely used in ovarian, breast and prostate cancer treatments, and are in clinical trials for many other cancers. Understanding how drugs are stored and distributed in cells could ultimately open the door to more personalised treatment strategies that improve success rates and reduce resistance or relapse.
Dr Louise Fets, senior author and Head of the LMS’ Drug Transport and Tumour metabolism Group, said: “By understanding how drugs are taken up into cells, we can understand whether this influences why cancer drugs work for some people and not for others. Eventually, we hope to be able study the molecular signature of a patient's tumour to help to tailor therapeutic approaches in a more personalised way."
This study used patient tumour tissue maintained outside the body. In real patients, drugs are delivered through the bloodstream, and tumour blood vessels are often disorganised, which may further increase uneven drug delivery. Future research will use animal models and larger patient studies to better understand how drug delivery, tumour structure and lysosomal storage interact in real clinical settings, including in relapsed cancers.
This research was supported by funding from the Medical Research Council, Cancer Research UK, a PhD studentship from the Integrative Toxicology Training Partnership administered by the MRC Toxicology Unit, and a Victoria’s Secret Global Fund for Women’s Cancers Career Development Award, in partnership with Pelotonia and AACR.
Infrastructure support for this research was provided by the NIHR Imperial Biomedical Research Centre (BRC).
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Article people, mentions and related links
Benjie Coleman
Faculty of Medicine
