Unlocking the Material Secrets Behind 20% Efficient Organic Solar Cells

New research has revealed how next-generation solar materials move energy with record breaking efficiency.
Organic solar cells use carbon-based molecules or polymers to absorb sunlight and generate electricity. These cells can be lightweight, flexible, adaptable and more sustainable than traditional silicon solar panels, and so are attracting global interest.
In just two decades, their efficiency has jumped from around 2% to more than 20% thanks to a new class of materials known as non-fullerene acceptors, particularly the high-performing “Y-family”.
Despite this rapid progress, it is not yet understood how these materials achieve such high efficiencies.po
In a new study published in Nature Materials, researchers led by Imperial College London are one step closer to uncovering this mystery, by identifying the molecular features that allow these next-generation materials to generate electricity so effectively.
Cracking the low-offset mystery
In traditional organic solar cells, two different materials – one donating electrons and one accepting them – must work together at a junction to split apart the tightly bound packets of light energy, known as excitons, into free electrical charges.
This normally requires a sizeable energy difference between the two materials, called a heterojunction, to force the charges apart. However, this can reduce the power the device produces.
The newest organic materials appear to break this rule. They achieve excellent performance with much smaller energy offsets, and some studies have even suggested that they could generate charges using a single material. This raises the possibility that the usual junction might not be necessary.
The team, led by Professor Jenny Nelson in Imperial’s Department of Physics, has now provided the most comprehensive explanation as to why this is. By combining measurements from real devices with a new computer model, the researchers were able to watch how excited states spread through these materials and influence energy conversion.
Comparing the model to experimental results they found that accounting for this state “spreading”, or delocalisation, is critical to understanding the remarkable efficiencies of these materials.
Lucy Hart, lead author and postdoctoral fellow at Imperial, said, “There has been a lot of debate about exactly how these exciting new materials generate electricity so efficiently when the traditional driving forces are so small.
“By combining our experimental measurements with a new computational model, we were able to pinpoint which molecular features drive this efficient charge generation.”
Co-author Daniel Medranda added, “Because these processes happen incredibly fast and at a molecular level in the materials, they are notoriously difficult to uncover. Our combined approach essentially acts as a highly advanced magnifying glass, allowing us to see exactly how the specific shape and packing of these molecules dictate the performance of the entire solar cell.”
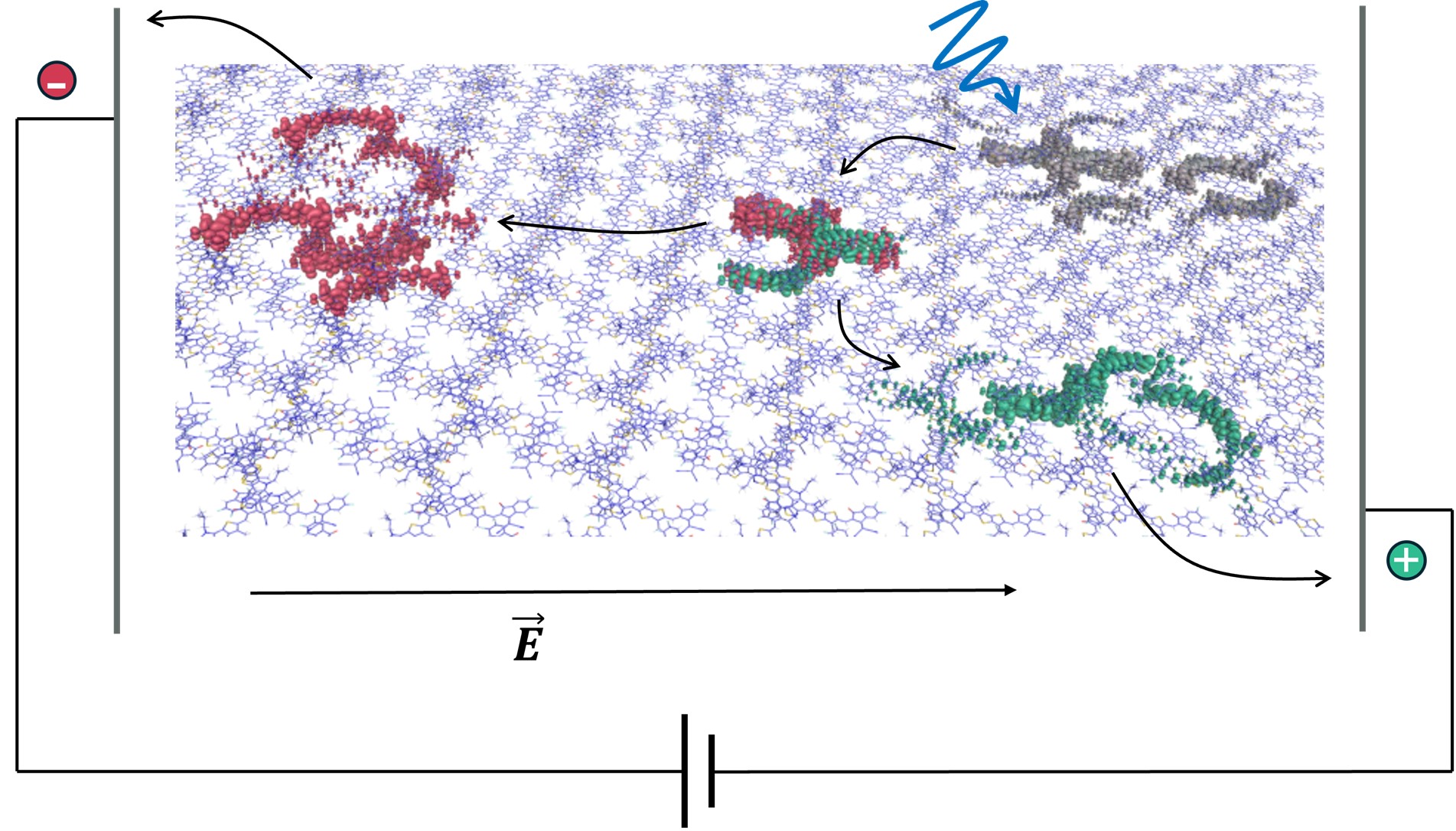
Visualisation of a photogenerated excited state (grey) evolving into separated positively (green) and negatively (red) charged regions of a molecular film. Credit: co-author Dr Daniel Medranda.
New rules for designing better solar materials
The study also identifies the key molecular features that enable such efficient charge generation.
The secret to their high efficiency lies in how the energy is shared across an entire molecular network. It’s this teamwork at the nanoscale that allows the charges to separate so effectively without needing a massive energetic push. Dr Flurin Eisner lecturer in green energy, Queen Mary University of London
Both the chemical structure of each molecule and the way the molecules are packed together are crucial. When both conditions align, the energy produced by light spreads more effectively across the material, helping charges separate without needing a large energetic “push”.
Dr Flurin Eisner, lecturer in green energy at Queen Mary University of London, said, “What our results make clear is that we can no longer look at these molecules in isolation. The secret to their high efficiency lies in how the energy is shared across an entire molecular network. It’s this teamwork at the nanoscale that allows the charges to separate so effectively without needing a massive energetic push.”
The team also tested whether the new materials could produce photocurrent entirely on their own, without the usual donor-acceptor junction. The answer, for now, is “not yet”, but the research clearly maps out how materials could be improved to move closer to this goal.
This work provides a set of clear design rules for chemists and engineers aiming to push organic solar cells to even higher efficiencies. The team suggests that future materials development should focus on reducing molecular reorganisation energies, minimising structural disorder and strengthening interactions between molecules.
The research was supported by UKRI (ATIP programme grant), the UKRI ERC underwrite scheme (POTENtIAl) and the Spanish CSIC, via collaboration with Professor Campoy-Quiles at ICMAB, Barcelona (project DOMMINO).
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Article people, mentions and related links
Eleanor Barrand
Faculty of Natural Sciences
