Aug 2025 - Article Published in Organic Chemistry Frontiers
by Alan Spivey
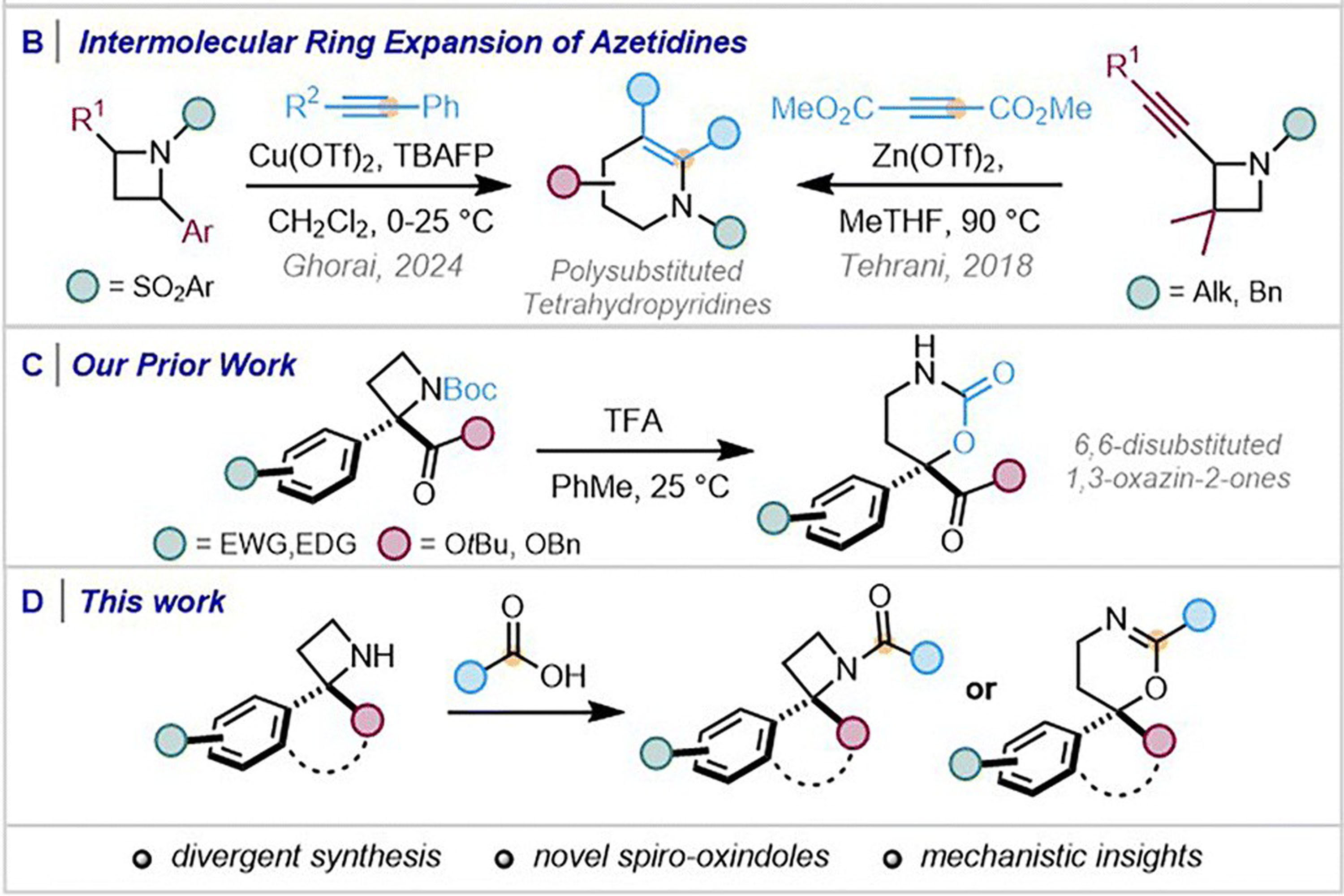
Adi Sahay’s work developing some new reactions of 2,2-disubstituted azetidines is published
- K. Sahay, C. S. Begg, X. Zhang, J. A. Bull and A. C. Spivey ‘Structurally divergent reactivity of 2,2-disubstituted azetidines – mechanistic insights and stereochemical implications of amide coupling and ring expansion to 5,6-dihydro-4H-1,3-oxazines’, 2025, 12, 6556-6563. DOI: https://doi.org/10.1039/D5QO00804B
Azetidines have gained traction in drug discovery for their ability to introduce conformational constraint and modulate physiochemical properties. Strategies that enable their selective functionalization or controlled expansion into more complex scaffolds provide opportunities for molecular diversification to rapidly access new chemical space. Subjecting 2,2-disubstituted azetidines to amide coupling with carboxylic acids is found to effect either N-acylation or ring expansion to spiro and 6,6-disubstituted 5,6-dihydro-4H-1,3-oxazine, dependent on reaction conditions. A diverse range of topologically interesting heterocycles, which hold significant potential for pharmaceutical screening, have been prepared using this divergent reaction manifold. A mechanistic framework, supported by additive screening and trapping experiments, is presented to account for the ring expansion and racemization that accompanies these transformations when the substrate allows formation of a ring-opened azafulvenium intermediate.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Article people, mentions and related links
Alan Spivey
Administration/Non-faculty departments
- Tel: 02075945841
- Email: a.c.spivey@imperial.ac.uk
- Articles by this author
