Dec 2025 - Article Published in Eu. J. Org. Chem.
by Alan Spivey
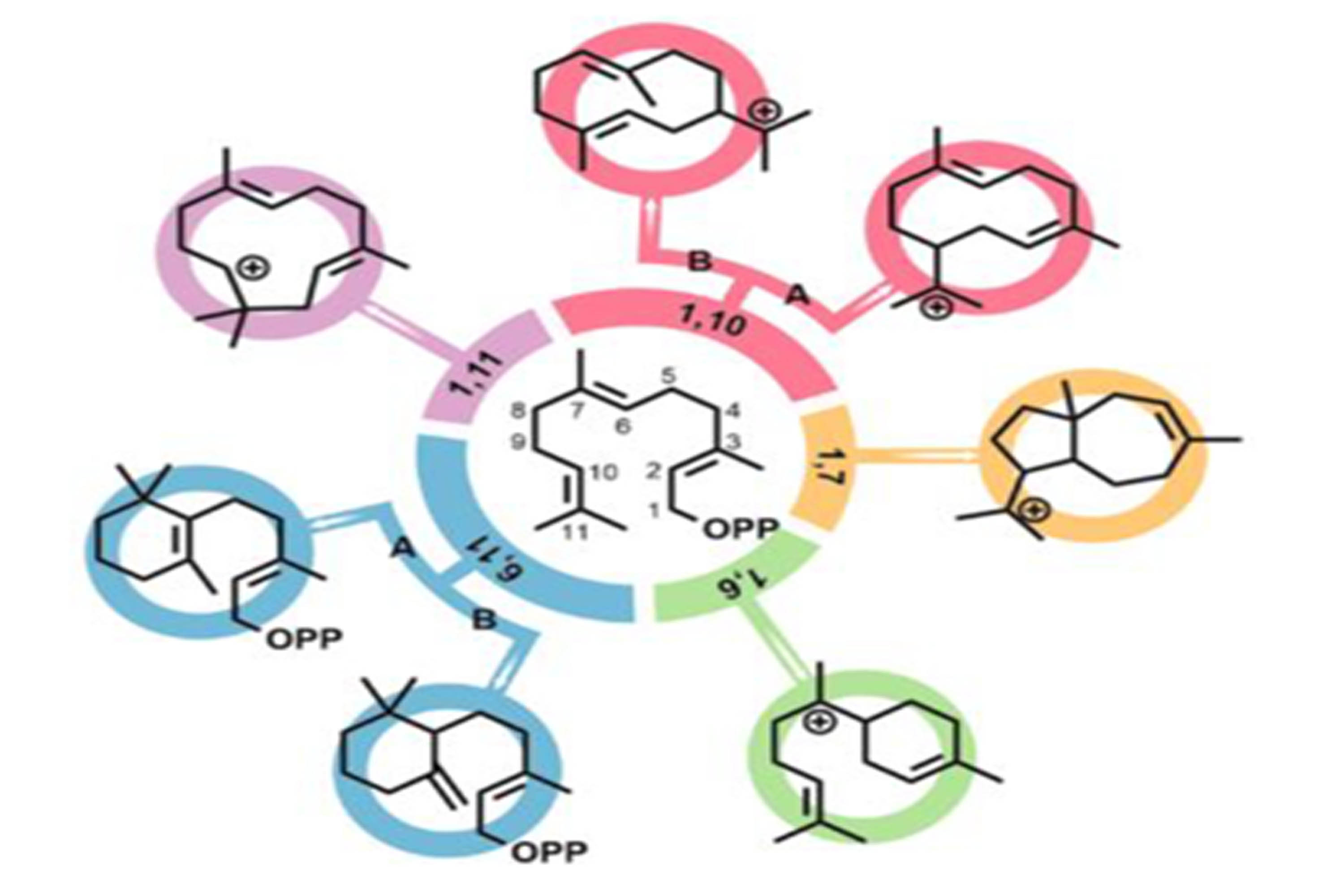
Chen Dai and Bohan Lin’s minireview discussing ring formation in sesquiterpenoid biomimetic and non-biomimetic synthesis is published.
- Lin, C. Dai and A. C. Spivey ‘Ring Formation in Sesquiterpene Synthesis: Biomimetic vs. Non-biomimetic Strategies’, 2025, 29, e202500816. DOI: http://doi.org/10.1002/ejoc.202500816
Biomimetic synthesis refers to organic synthesis using strategies that mimic biosynthetic pathways to prepare natural products. It draws inspiration from nature's efficiency in constructing organic compounds, often enabling mild conditions and efficient routes. Natural products constitute an enduring source of leads for drug discovery, and this approach has been used to successfully synthesise many medicinally important compounds. The prevalence of topologically complex carbocyclic ring-systems in many secondary metabolites and particularly terpenes, makes their preparation challenging and highlights the need for increased synthetic efficiency in carbocyclisation reactions. Here, we briefly review recent advances in our understanding of sesquiterpene cyclases and then present six case-studies comparing biomimetic vs. non-biomimetic approaches to the key ring-forming reactions in this context. Examples have been selected to exemplify some of the classic ring-systems that characterise this iconic natural product family.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Article people, mentions and related links
Alan Spivey
Administration/Non-faculty departments
- Tel: 02075945841
- Email: a.c.spivey@imperial.ac.uk
- Articles by this author
