Results
- Showing results for:
- Reset all filters
Search results
-
Journal articleOuyang L, Armstrong J, Chen Q, et al., 2020,
Void-free 3D bioprinting for in-situ endothelialization and microfluidic perfusion
, Advanced Functional Materials, Vol: 30, ISSN: 1616-301XTwo major challenges of 3D bioprinting are the retention of structural fidelity and efficient endothelialization for tissue vascularization. We address both of these issues by introducinga versatile3D bioprinting strategy, in which a templating bioink is deposited layer-by-layer alongside a matrix bioink to establish void-free multimaterial structures. After crosslinking the matrix phase, the templating phase issacrificedto create a well-defined 3D network of interconnected tubular channels. This void-free 3D printing (VF-3DP) approachcircumvents the traditional concerns of structural collapse, deformation and oxygen inhibition, moreover, it can be readily used to printmaterials that are widely considered “unprintable”. By pre-loading endothelial cells into the templating bioink, the inner surface of the channels can be efficiently cellularized with a confluent endothelial layer. This in-situ endothelializationmethod can be used to produce endothelium with a far greater uniformity than can be achieved using the conventional post-seeding approach. This VF-3DP approach canalsobe extended beyond tissue fabrication and towards customized hydrogel-based microfluidics and self-supported perfusable hydrogel constructs.
-
Journal articleJabbour RJ, Owen TJ, Pandey P, et al., 2020,
Future potential of engineered heart tissue patches for repairing the damage caused by heart attacks
, EXPERT REVIEW OF MEDICAL DEVICES, Vol: 17, Pages: 1-3, ISSN: 1743-4440- Author Web Link
- Cite
- Citations: 7
-
Journal articleHong SP, Lombardo Y, Chan TE, et al., 2019,
Single-cell transcriptomics reveals multi-step adaptations to endocrine therapy
, Nature Communications, Vol: 10, ISSN: 2041-1723Resistant tumours are thought to arise from the action of Darwinian selection on genetically heterogenous cancer cell populations. However, simple clonal selection is inadequate to describe the late relapses often characterising luminal breast cancers treated with endocrine therapy (ET), suggesting a more complex interplay between genetic and non-genetic factors. Here, we dissect the contributions of clonal genetic diversity and transcriptional plasticity during the early and late phases of ET at single-cell resolution. Using single-cell RNA-sequencing and imaging we disentangle the transcriptional variability of plastic cells and define a rare sub population of pre-adapted (PA) cells which undergoes further transcriptomic reprogramming and copy number changes to acquire full resistance. We find evidence for sub-clonal expression of a PA signature in primary tumours and for dominant expression in clustered circulating tumour cells. We propose a multi-step model for ET resistance development and advocate the use of stage-specific biomarkers.
-
Journal articleBaxan N, Papanikolaou A, Salles-Crawley I, et al., 2019,
Characterization of acute TLR-7 agonist-induced hemorrhagic myocarditis in mice by multiparametric quantitative cardiac magnetic resonance imaging.
, Dis Model Mech, Vol: 12Hemorrhagic myocarditis is a potentially fatal complication of excessive levels of systemic inflammation. It has been reported in viral infection, but is also possible in systemic autoimmunity. Epicutaneous treatment of mice with the Toll-like receptor 7 (TLR-7) agonist Resiquimod induces auto-antibodies and systemic tissue damage, including in the heart, and is used as an inducible mouse model of systemic lupus erythematosus (SLE). Here, we show that overactivation of the TLR-7 pathway of viral recognition by Resiquimod treatment of CFN mice induces severe thrombocytopenia and internal bleeding, which manifests most prominently as hemorrhagic myocarditis. We optimized a cardiac magnetic resonance (CMR) tissue mapping approach for the in vivo detection of diffuse infiltration, fibrosis and hemorrhages using a combination of T1, T2 and T2* relaxation times, and compared results with ex vivo histopathology of cardiac sections corresponding to CMR tissue maps. This allowed detailed correlation between in vivo CMR parameters and ex vivo histopathology, and confirmed the need to include T2* measurements to detect tissue iron for accurate interpretation of pathology associated with CMR parameter changes. In summary, we provide detailed histological and in vivo imaging-based characterization of acute hemorrhagic myocarditis as an acute cardiac complication in the mouse model of Resiquimod-induced SLE, and a refined CMR protocol to allow non-invasive longitudinal in vivo studies of heart involvement in acute inflammation. We propose that adding T2* mapping to CMR protocols for myocarditis diagnosis improves diagnostic sensitivity and interpretation of disease mechanisms.This article has an associated First Person interview with the first author of the paper.
-
Journal articleGopal S, Chiappini C, Armstrong J, et al., 2019,
Immunogold FIB-SEM: combining volumetric ultrastructure visualization with 3D biomolecular analysis to dissect cell-environment interactions
, Advanced Materials, Vol: 31, Pages: 1-8, ISSN: 0935-9648Volumetric imaging techniques capable of correlating structural and functional information with nanoscale resolution are necessary to broaden the insight into cellular processes within complex biological systems. The recent emergence of focused ion beam scanning electron microscopy (FIB‐SEM) has provided unparalleled insight through the volumetric investigation of ultrastructure; however, it does not provide biomolecular information at equivalent resolution. Here, immunogold FIB‐SEM, which combines antigen labeling with in situ FIB‐SEM imaging, is developed in order to spatially map ultrastructural and biomolecular information simultaneously. This method is applied to investigate two different cell–material systems: the localization of histone epigenetic modifications in neural stem cells cultured on microstructured substrates and the distribution of nuclear pore complexes in myoblasts differentiated on a soft hydrogel surface. Immunogold FIB‐SEM offers the potential for broad applicability to correlate structure and function with nanoscale resolution when addressing questions across cell biology, biomaterials, and regenerative medicine.
-
Journal articleBowling S, Lawlor K, Rodriguez T, 2019,
Cell competition: The winners and losers of fitness selection
, Development, Vol: 146, Pages: 1-12, ISSN: 0950-1991The process of cell competition results in the elimination of cells that are viable but “less fit” than surrounding cells. Given the highly heterogeneous nature of our tissues, it seems increasingly likely that cells are engaged in a “survival of the fittest” battle throughout life. The process has myriad positive roles in the organism: it selects against mutant cells in developing tissues, prevents the propagation of oncogenic cells, and eliminates damaged cells during ageing. However, “super-fit” cancer cells can exploit cell competition mechanisms to expand and spread. Here, we review the regulation, roles and risks of cell competition in organism development, ageing and disease.
-
Journal articleOwen TJ, Harding SE, 2019,
Multi-cellularity in cardiac tissue engineering, how close are we to native heart tissue?
, Journal of Muscle Research and Cell Motility, Vol: 40, Pages: 151-157, ISSN: 0142-4319Tissue engineering is a complex field where the elements of biology and engineering are combined in an attempt to recapitulate the native environment of the body. Tissue engineering has shown one thing categorically; that the human body is extremely complex and it is truly a difficult task to generate this in the lab. There have been varied attempts at trying to generate a model for the heart with numerous cell types and different scaffolds or materials. The common underlying theme in these approaches is to combine together matrix material and different cell types to make something similar to heart tissue. Multi-cellularity is an essential aspect of the heart and therefore critical to any approach which would try to mimic such a complex tissue. The heart is made up of many cell types that combine to form complex structures like: deformable chambers, a tri-layered heart muscle, and vessels. Thus, in this review we will summarise how tissue engineering has progressed in modelling the heart and what gaps still exist in this dynamic field.
-
Journal articleWatson S, Duff J, Bardi I, et al., 2019,
Biomimetic electromechanical stimulation to maintain adult myocardial slices in vitro
, Nature Communications, Vol: 10, ISSN: 2041-1723Adult cardiac tissue undergoes a rapid process of dedifferentiation when cultured outside the body. The in vivo environment, particularly constant electromechanical stimulation, is fundamental to the regulation of cardiac structure and function. We investigated the role of electromechanical stimulation in preventing culture-induced dedifferentiation of adult cardiac tissue using rat, rabbit and human heart failure myocardial slices. Here we report that the application of a preload equivalent to sarcomere length (SL) = 2.2 μm is optimal for the maintenance of rat myocardial slice structural, functional and transcriptional properties at 24 h. Gene sets associated with the preservation of structure and function are activated, while gene sets involved in dedifferentiation are suppressed. The maximum contractility of human heart failure myocardial slices at 24 h is also optimally maintained at SL = 2.2 μm. Rabbit myocardial slices cultured at SL = 2.2 μm remain stable for 5 days. This approach substantially prolongs the culture of adult cardiac tissue in vitro.
-
Journal articleHellen N, Pinto Ricardo C, Vauchez K, et al., 2019,
Proteomic analysis reveals temporal changes in protein expression in human induced pluripotent stem cell-derived cardiomyocytes in vitro
, Stem Cells and Development, Vol: 28, ISSN: 1547-3287Human induced pluripotent stem cell-derived cardiomyocytes hold great promise for regenerative medicine and in vitro screening. Despite displaying key cardiomyocyte phenotypic characteristics, they more closely resemble foetal/neonatal cardiomyocytes and further characterisation is necessary. Combining the use of tandem mass tags to label cell lysates, followed by multiplexing, we have determined the effects of short term (30 day) in vitro culture on human induced pluripotent stem cell derived cardiomyocyte protein expression. We found that human induced pluripotent stem cell derived cardiomyocytes exhibit temporal changes in global protein expression; alterations in protein expression were pronounced during the first 2 weeks following thaw and dominated by reductions in proteins associated with protein synthesis and ubiquitination. Between 2 and 4 weeks proceeding thaw alterations in protein expression were dominated by metabolic pathways, indicating a potential temporal metabolic shift from glycolysis towards oxidative phosphorylation. Time-dependent changes in proteins associated with cardiomyocyte contraction, excitation-contraction coupling and metabolism were detected. While some were associated with expected functional outcomes in terms of morphology or electrophysiology, others such as metabolism did not produce the anticipated maturation of human induced pluripotent stem cell derived cardiomyocytes. In several cases, a predicted outcome was not clear because of the concerted changes in both stimulatory and inhibitory pathways. Nevertheless, clear development of human induced pluripotent stem cell derived cardiomyocytes over this time period was evident.
-
Journal articleLi C, Ouyang L, Pence I, et al., 2019,
Buoyancy-driven gradients for biomaterial fabrication and tissue engineering
, Advanced Materials, Vol: 31, ISSN: 0935-9648The controlled fabrication of gradient materials is becoming increasingly important as the next generation of tissue engineering seeks to produce inhomogeneous constructs with physiological complexity. Current strategies for fabricating gradient materials can require highly specialized materials or equipment and cannot be generally applied to the wide range of systems used for tissue engineering. Here, the fundamental physical principle of buoyancy is exploited as a generalized approach for generating materials bearing well‐defined compositional, mechanical, or biochemical gradients. Gradient formation is demonstrated across a range of different materials (e.g., polymers and hydrogels) and cargos (e.g., liposomes, nanoparticles, extracellular vesicles, macromolecules, and small molecules). As well as providing versatility, this buoyancy‐driven gradient approach also offers speed (<1 min) and simplicity (a single injection) using standard laboratory apparatus. Moreover, this technique is readily applied to a major target in complex tissue engineering: the osteochondral interface. A bone morphogenetic protein 2 gradient, presented across a gelatin methacryloyl hydrogel laden with human mesenchymal stem cells, is used to locally stimulate osteogenesis and mineralization in order to produce integrated osteochondral tissue constructs. The versatility and accessibility of this fabrication platform should ensure widespread applicability and provide opportunities to generate other gradient materials or interfacial tissues.
This data is extracted from the Web of Science and reproduced under a licence from Thomson Reuters. You may not copy or re-distribute this data in whole or in part without the written consent of the Science business of Thomson Reuters.
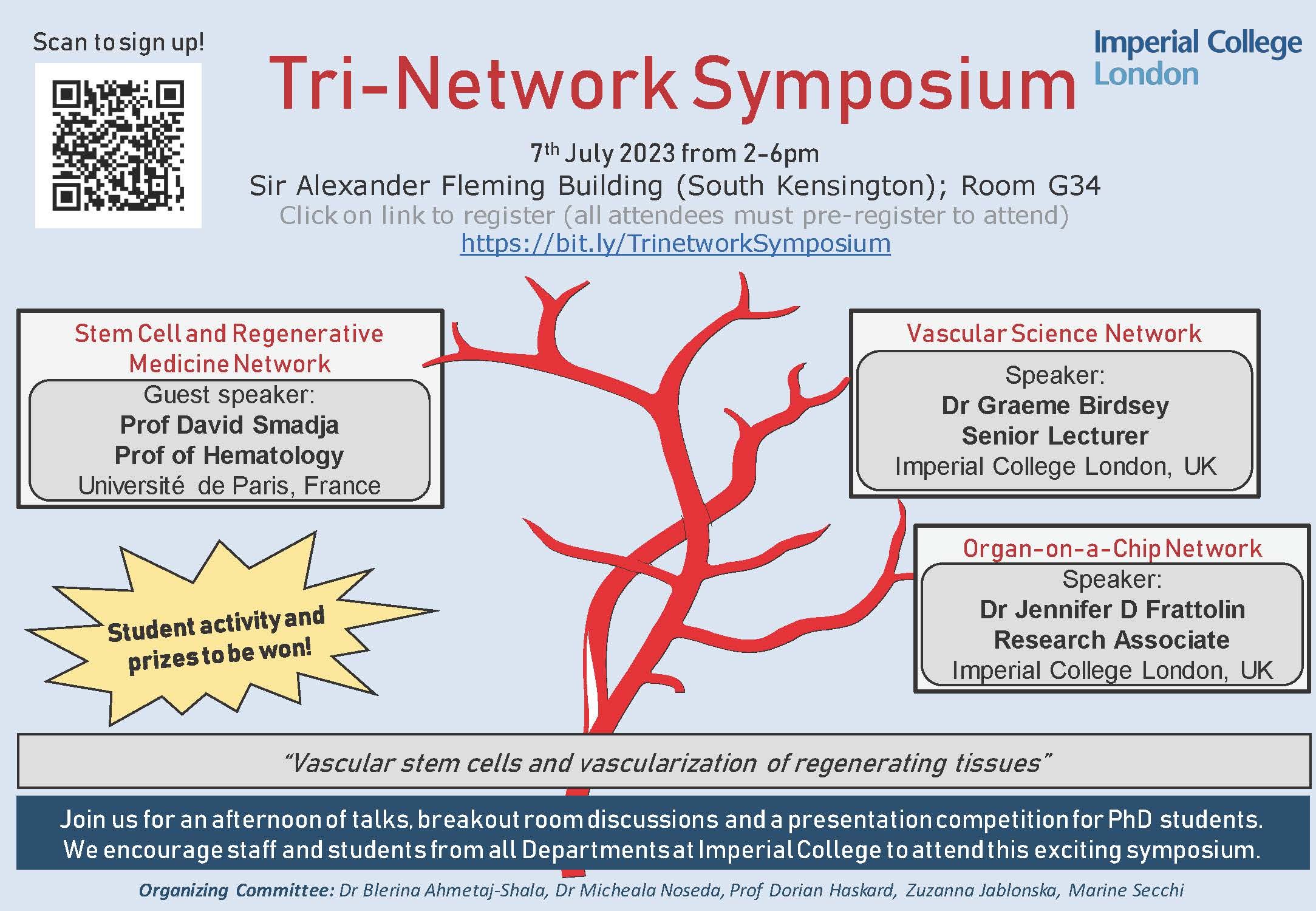
Join our network
If you would like to become a member of the network please contact us.
- Sign-up
- Read about member benefits
Coordination and contact
Network Lead
Dr Michela Noseda
Network Lead
Dr Blerina Ahmetaj-Shala
Network Coordinator
Zuzanna Jablonska
Network Coordinator
Marine Secchi
General enquiries
ISRMNcontact@imperial.ac.uk