Bio-inspired Artificial Pancreas
Research Team: Dr Pantelis Georgiou, Dr Pau Herrero, Prof Nick Oliver, Dr Monika Reddy, Mr Ryan Armiger, Mr John Daniels, Dr Mohamed El Sharkawy, Dr Peter Pesl, Dr Shivshankar Seechurn, Dr Chukwuma Uduku, Ms Narvada Jugnee, Prof Des Johnston, and Prof Chris Toumazou
Funding: The Wellcome Trust; Imperial College London Diabetes Centre (Abu Dhabi, UAE)
Technological progress in the field of diabetes management has enabled the development of automated insulin delivery systems for blood glucose control, the so-called artificial pancreas, which consists of a subcutaneous continuous glucose sensor and a subcutaneous infusion pump that delivers insulin at a rate decided by a computer program.
A bio-inspired approach: We have introduced the Bio-inspired Artificial Pancreas (BiAP), the first and only closed-loop insulin delivery system based on mathematical modelling of the pancreatic beta-cell physiology.
An AI-powered insulin bolus calculator: Due to pharmacological limitations of currently available insulin formulations, artificial pancreas systems require user intervention to administer an additional insulin dose at mealtimes. For this purpose, BiAP incorporates an innovative meal-insulin bolus calculator which uses artificial intelligence to learn from user and controller behaviours in order to provide superior meal-insulin dosing.
A low-power solution: The BiAP control algorithm is designed for embedded low-power solutions (e.g. insulin pumps) and has successfully been implemented in a dedicated microchip-based solution and, more recently, in a smartphone-based platform. It has also been integrated with different commercial insulin pumps and continuous glucose sensors.
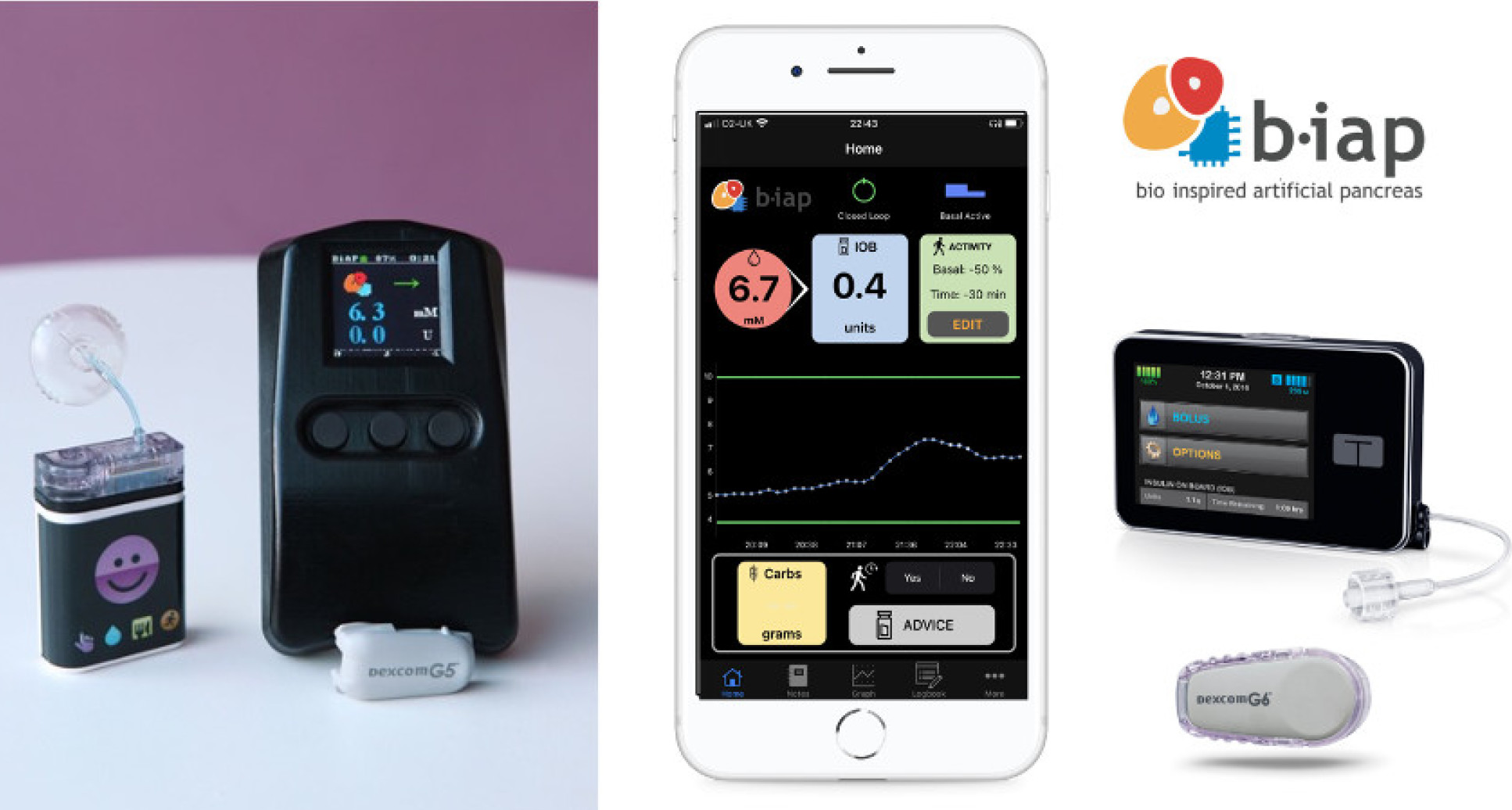
Figure. The Bio-inspired Artificial Pancreas. Left) Microchip-based handheld version; Right) Smartphone-based version
Clinical trials:
BiAP has been clinically evaluated in participants with type 1 diabetes in the following trials:
- Feasibility studies including fasting conditions, overnight and following a standard meal (breakfast) challenge (n=20)
- 24-hour randomised controlled clinical trial including unannounced meals and underestimated carbohydrate content (n=12)
- 24-hour randomised controlled clinical trial including standard exercise with insulin-only closed-loop control and insulin and glucagon closed-loop control (n=6)
- 3-month crossover randomised controlled clinical trial in ambulatory conditions (n=20) (trial on-hold)
Follow us on Twitter: @Imperial_BiAP
BiAP being presented at the MedTech SuperConnector and Techcelerate Showcase 2021
Related Publications:
- Herrero, P., El-Sharkawy, M., Daniels, J., Jugnee, N., Uduku, C. N., Reddy, M., Oliver, N. & Georgiou, P. The bio-inspired artificial pancreas for type 1 diabetes control in the home: system architecture and preliminary results. Journal of diabetes science and technology, 2019, 13, 1017–1025.
- Herrero, P., Bondia, J., Adewuyi, O., Pesl, P., El-Sharkawy, M., Reddy, M., Toumazou, C., Oliver, N & Georgiou, P. (2017). Enhancing automatic closed-loop glucose control in type 1 diabetes with an adaptive meal bolus calculator–in silico evaluation under intra-day variability. Computer methods and programs in biomedicine, 146, 125-131.
- Herrero, P., Bondia, J., Oliver, N. & Georgiou, P. (2017) A coordinated control strategy for insulin and glucagon delivery in type 1 diabetes. Computer methods in biomechanics and biomedical engineering. 20,1474–1482.x
- Seechurn S, Reddy M, Jugnee N, El Sharkawy M, Pesl P, Herrero P, Godsland I, Toumazou C, Georgiou P, Oliver N. Does the addition of glucagon to a closed loop system impact on post exercise glycaemia? Diabetes Technology and Therapeutics 2016;18(S1):A148
- Seechurn S, Reddy M, Jugnee N, El Sharkawy M, Pesl P, Herrero P, Godsland I, Toumazou C, Georgiou P, Oliver N. Does the addition of glucagon to the closed loop insulin pump add any benefit? Diabetes Technology and Therapeutics 2016;18(S1):A149
- Reddy, M., Herrero, P., Sharkawy, M. E., Pesl, P., Jugnee, N., Pavitt, D., Pavitt, D., Godsland, I. F., Alberti, G., Toumazou, C., Johnston, D. G., Georgiou, P., & Oliver, N. S. (2016). Metabolic control with the bio-inspired artificial pancreas in adults with type 1 diabetes: a 24-hour randomized controlled crossover study. Journal of diabetes science and technology, 10(2), 405-413.
- Reddy, M., Herrero, P., El Sharkawy, M., Pesl, P., Jugnee, N., Thomson, H., Pavitt, D., Toumazou, C.,Johnston, D., Georgiou, P. & Oliver, N. (2014). Feasibility study of a bio-inspired artificial pancreas in adults with type 1 diabetes. Diabetes technology & therapeutics, 16(9), 550-557.
- Herrero P, Georgiou P, Oliver N, et al, A Bio-Inspired Glucose Controller Based on the Pancreatic beta-Cell Physiology, Journal of Diabetes Science and Technology, 2012, Vol:6
- Oliver N, Georgiou P, Johnston D, et al, A benchtop closed-loop system controlled by a bio-inspired silicon implementation of the pancreatic beta cell., J Diabetes Sci Technol, 2009, Vol:3, Pages:1419-1424
- Georgiou P, Toumazou C. A Silicon Pancreatic Beta Cell for Diabetes, IEEE Transactions on Biomedical Circuits and Systems, vol.1, no.1, pp.39-49, March 2007
- Georgiou P, Toumazou C. Towards an ultra low power chemically inspired electronic beta cell for diabetes, 2006 IEEE International Symposium on Circuits and Systems. ISCAS 2006, Proceedings, pp. 4, 21-24 May 2006



