Latest network news and research results
- Single-Molecule Binding Assay Using Nanopores and Dimeric NP Conjugates. Advanced Materials 2021

The ability to measure biomarkers, both specifically and selectively at the single-molecule level in biological fluids, has the potential to transform the diagnosis, monitoring, and therapeutic intervention of diseases. The use of nanopores has been gaining prominence in this area, not only for sequencing but more recently in screening applications. The selectivity of nanopore sensing can be substantially improved with the use of labels, but substantial challenges remain, especially when trying to differentiate between bound from unbound targets. Here highly sensitive and selective molecular probes made from nanoparticles (NPs) that self-assemble and dimerize upon binding to a biological target are designed. It is shown that both single and paired NPs can be successfully resolved and detected at the single-molecule nanopore sensing and can be used for applications such as antigen/antibody detection and microRNA (miRNA) sequence analysis. It is expected that such technology will contribute significantly to developing highly sensitive and selective strategies for the diagnosis and screening of diseases without the need for sample processing or amplification while requiring minimal sample volume.
- Single-molecule amplification-free multiplexed detection of circulating microRNA cancer biomarkers from serum. Nature Communications 2021
MicroRNAs (miRNAs) play essential roles in post-transcriptional gene expression and are also found freely circulating in bodily fluids such as blood. Dysregulated miRNA signatures have been associated with many diseases including cancer, and miRNA profiling from liquid biopsies offers a promising strategy for cancer diagnosis, prognosis and monitoring. Here, we develop size-encoded molecular probes that can be used for simultaneous electro-optical nanopore sensing of miRNAs, allowing for ultrasensitive, sequence-specific and multiplexed detection directly in unprocessed human serum, in sample volumes as small as 0.1 μl. We show that this approach allows for femtomolar sensitivity and single-base mismatch selectivity. We demonstrate the ability to simultaneously monitor miRNAs (miR-141-3p and miR-375-3p) from prostate cancer patients with active disease and in remission. This technology can pave the way for next generation of minimally invasive diagnostic and companion diagnostic tests for cancer.
- Combined quantum tunnelling and dielectrophoretic trapping for molecular analysis at ultra-low analyte concentrations. Nature Communications 2021
Quantum tunnelling offers a unique opportunity to study nanoscale objects with atomic resolution using electrical readout. However, practical implementation is impeded by the lack of simple, stable probes, that are required for successful operation. Existing platforms offer low throughput and operate in a limited range of analyte concentrations, as there is no active control to transport molecules to the sensor. We report on a standalone tunnelling probe based on double-barrelled capillary nanoelectrodes that do not require a conductive substrate to operate unlike other techniques, such as scanning tunnelling microscopy. These probes can be used to efficiently operate in solution environments and detect single molecules, including mononucleotides, oligonucleotides, and proteins. The probes are simple to fabricate, exhibit remarkable stability, and can be combined with dielectrophoretic trapping, enabling active analyte transport to the tunnelling sensor. The latter allows for up to 5-orders of magnitude increase in event detection rates and sub-femtomolar sensitivity.
- Monitoring amyloid-β 42 conformational change using a spray-printed graphene electrode. Paper available here.
Up to now, the reproducibility and stability of graphene-based electrochemical sensors have represented an obstacle to the development of practical biosensing techniques. In this paper we report a cost-effective and highly reproducible graphene-based electrochemical sensing platform to monitor the kinetic conformational change of amyloidogenic proteins. The sensor surface is spray-printed with a graphene oxide layer and then electrochemically reduced to achieve excellent sensitivity to the redox current. The reproducibility of these sensors in terms of redox peak position, intensity and electroactive area has been proved to be high. These sensors are used to monitor the conformational changes of amyloid-β 42 via the change in the oxidation current of tyrosine, which is caused by different electrochemical accessibility during the aggregation process. The aggregation process detected at these graphene electrochemical sensors shows a good correlation with the fluorescence assay. The proposed platform provides a complementary technique to aid understanding of the detailed process of amyloidogenic protein aggregation and the mechanism of neurodegenerative diseases as well as helping to promote the development of disease-prevention strategies.
- Professor Norbert Klein introduces graphene-based COVID-19 detection to ISST Innvoation Ecosystem
Read the article based on Norbert's talk here.
- Carbon-Dot-Enhanced Graphene Field-Effect Transistors for Ultrasensitive Detection of Exosomes, ACS Appl. Mater. Interfaces 2021
Graphene field-effect transistors (GFETs) are suitable building blocks for high-performance electrical biosensors, because graphene inherently exhibits a strong response to charged biomolecules on its surface. However, achieving ultralow limit-of-detection (LoD) is limited by sensor response time and screening effect. Herein, we demonstrate that the detection limit of GFET biosensors can be improved significantly by decorating the uncovered graphene sensor area with carbon dots (CDs). The developed CDs-GFET biosensors used for exosome detection exhibited higher sensitivity, faster response, and three orders of magnitude improvements in the LoD compared with nondecorated GFET biosensors. A LoD down to 100 particles/μL was achieved with CDs-GFET sensor for exosome detection with the capability for further improvements. The results were further supported by atomic force microscopy (AFM) and fluorescent microscopy measurements. The high-performance CDs-GFET biosensors will aid the development of an ultrahigh sensitivity biosensing platform based on graphene for rapid and early diagnosis of diseases.
- Yi-Lun Ying, Aleksandar P. Ivanov (Chemistry, Imperial College) and Vincent Tabard-Cossa report on recent developments discussed at the 2020 Nanopore Electrochemistry Meeting. Nature Chemistry 2021
-
Facile biosensors for rapid detection of COVID-19
Dr Lizhou Xu from Professor Klein's lab has written a review on COVID 19 detection which is now published in Biosensor and Bioelectronics. Access the paper here.
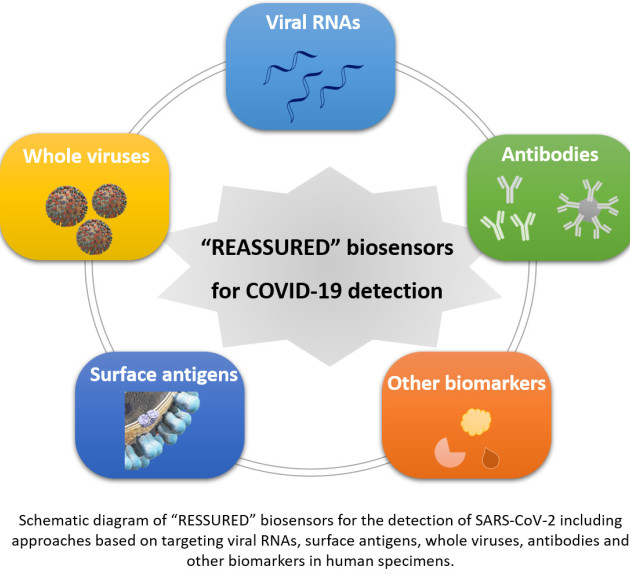
Abstract:
Currently the world is being challenged by a public health emergency caused by the coronavirus pandemic (COVID-19). Extensive efforts in testing for coronavirus infection, combined with isolating infected cases and quarantining those in contact, have proven successful in bringing the epidemic under control. Rapid and facile screening of this disease is in high demand. This review summarises recent advances in strategies reported by international researchers and engineers concerning how to tackle COVID-19 via rapid testing, mainly through nucleic acid- and antibody- testing. The roles of biosensors as powerful analytical tools are emphasized for the detection of viral RNAs, surface antigens, whole viral particles, antibodies and other potential biomarkers in human specimen. We critically review in depth newly developed biosensing methods especially for in-field and point-of-care detection of SARS-CoV-2. Additionally, this review describes possible future strategies for virus rapid detection. It helps researchers working on novel sensor technologies to tailor their technologies in a way to address the challenge for effective detection of COVID-19.