Neural correlates of sensorimotor control in neonates
Preterm babies born with 5 or 6 months have a relatively high risk of developing cerebral palsy (CP), a permanent neurological disorder affecting the control of movement and posture. In order to find a cure for CP it is important to understand the mechanisms underlying motor control in neonates. Furthermore, it is critical to understand the brain development in infants in relation to sensorimotor language acquisition.
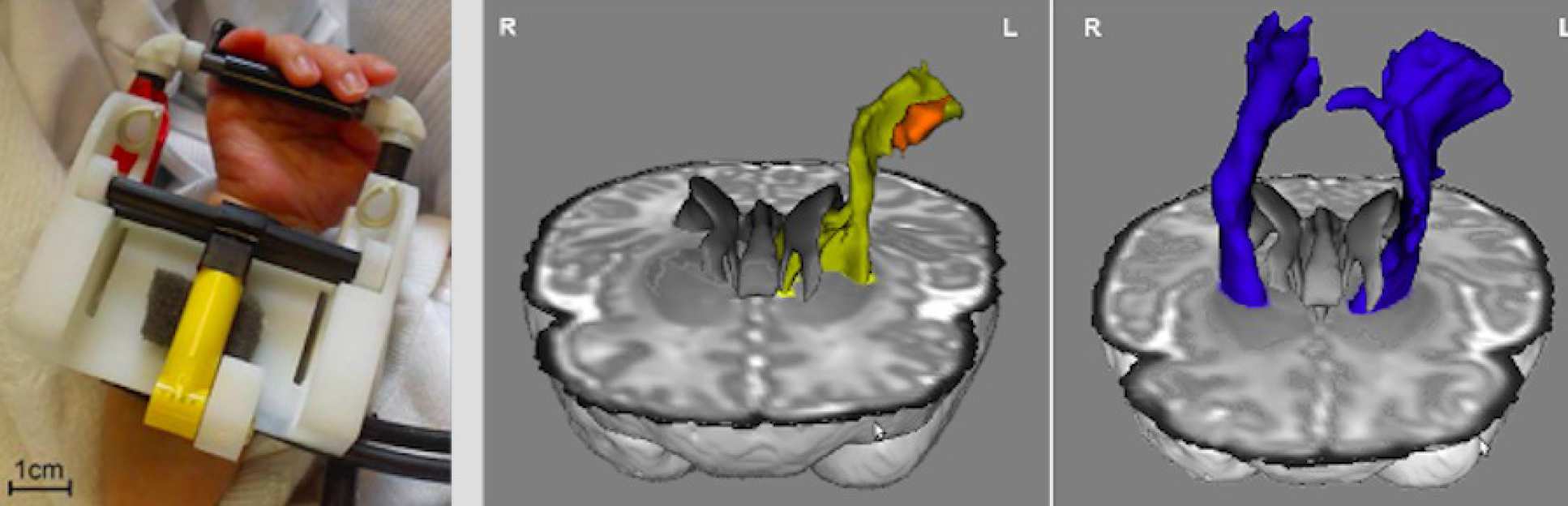
We develop tiny soft robotic devices to control the limbs movement of infant lying inside a magnetic resonance imaging (MRI) scanner. Using functional MRI (fMRI) and diffusion tensor MRI (DTI), this enables us to investigate the brain activity and connectivity during well controlled movement. This research is done with Tomoki Arichi, David Edwards and other members of the Centre for the Developing Brain at Kings College London https://www.developingbrain.co.uk.
First, we found that the hemodynamic response function (HRF) characterising the activation MRI dynamics varies significantly varies from preterm to term infants and from infants to adults. We derived the HRF that should be used to analyse infants data, as using the adults HRF would lead to erroneous results.
Later, we elucidated how the sensory-motor activity develops in the last trimester before term equivalent age. The functional responses become more complex as infants mature, with an involvement of accessory areas and the ipsilateral hemisphere, likely associated with increasing growth of structural connections. The overall response decreases is observed near term.
fMRI and DTI enabled us to analyse the alterations of structural and functional connectivity in the sensorimotor system of infants with perinatal brain injury. A marked asymmetry was observed in the efferent corticospinal tracts, with a decreased volume in the lesional hemisphere. Altered trajectories circumvent the periventricular white matter lesion to meet the fMRI clusters.
More recently, we have observed the cortical motor activity at 2/3 of the gestation, which already exhibits a cortical organisation similar to adults. This means that if a brain injury is happening after this time, the recovery will be determined/affected by this organisation. We have also used our tools to investigate the basic mechanism of the associative learning in newborns (Cerebral Cortex 2020).
Some related publications
- T Arichi, A Moraux, A Melendez, V Doria, M Groppo, N Merchant, S Combs, E Burdet, DJ Larkman, SJ Counsell, CF Beckmann and AD Edwards (2010), Somatosensory cortical activation identified by functional MRI in preterm and term infants. Neuroimage 49(3): 2063-71.
- T Arichi, G Fagiolo, M Varela, A Melendez-Calderon, AG Allievi, N Merchant, N Tusor, SJ Counsell, E Burdet, CF Beckmann and AD Edwards (2012), Development of BOLD signal hemodynamic responses in the human brain. Neuroimage 63(2): 663-73.
- T Arichi, SJ Counsell, AG Allievi, AT Chew, M Martinez-Biarge, V Mondi, N Tusor, N Merchant, E Burdet, FM Cowan and AD Edwards (2014), The effects of hemorrhagic parenchymal infarction on the establishment of sensorimotor structural and functional connectivity in early infancy. Neuroradiology 56: 985-94, 2014.
- AG Allievi, T Arichi, AL Gordon and E Burdet (2014), Technology-aided assessment of motor function in early infancy. Frontiers in Neurology, https://doi.org/10.3389/fneur.2014.00197
- A Allievi, T Arichi, AD Edwards and E Burdet (2015), Maturation of sensori-motor functional responses during the third trimester of human development. Cerebral Cortex 26 (1): 402-13
- S DallOrso, A Allievi, J Steinweg, D Edwards, E Burdet, T Arichi (2018), Somatotopic mapping of the developing sensorimotor cortex in the preterm human brain. Cerebral Cortex 28(7): 2507-15.
- S Dall’Orso, WP Fifer, PD Balsam, J Brandon, C O’Keefe, T Poppe, K Vecchiato, AD Edwards, E Burdet and T Arichi (2020), Cortical processing of multi-modal sensory learning in human neonates. Cerebral Cortex 31(3): 1827-36.