For polio, two vaccines are better than one
by Sam Wong
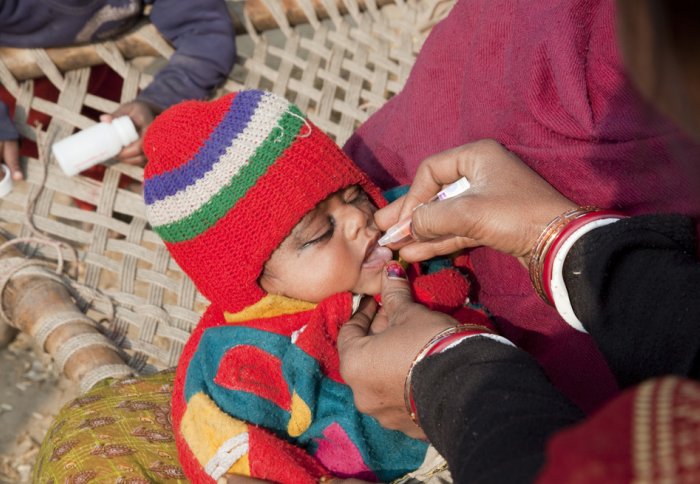
A child receives oral polio vaccine drops from a house-to-house polio vaccination team in Bihar, India. Photo: Gates Foundation/Flickr
Using both live and inactivated polio vaccines could help speed the global eradication of polio, a new study reports.
The study, done in Indian children already given the live polio vaccine, shows that a single dose of the inactivated vaccine boosts immunity more effectively than an additional live vaccine dose.
The results, published this week in Science, have already contributed to a decision by World Health Organisation (WHO) advisers to use the inactivated vaccine more widely in the push to eradicate polio.
In certain parts of the globe, like Pakistan and Nigeria, polio is proving difficult to eradicate. Making the choice between an oral live attenuated vaccine (OPV) and an injected inactivated vaccine (IPV) has been highly controversial.
To date, OPV has been more widely used because it is cheaper, requires no needles to administer, and is better able to induce immunity in the intestinal lining, known as mucosal immunity. However, the immune response to OPV is highly variable and mucosal immunity appears to diminish rapidly after OPV, meaning that several doses of this vaccine may be required, a difficult feat in remote or conflict zones.
Professor Nick Grassly from the School of Public Health at Imperial College London, who co-authored the study, said: “The decision to use inactivated poliovirus vaccine was previously controversial because it was unclear whether it would improve intestinal immunity. But in the last year we’ve published two studies that have provided convincing evidence that when given to children who previously received oral vaccine, the inactivated vaccine offers a very effective boost. This means that both OPV and IPV have an important role to play in the eradication programme. The results have already fed into the WHO’s eradication strategy, and campaigns with the vaccine have started in Nigeria and are planned for Pakistan, two countries with persistent poliovirus transmission.”
Researchers led by the WHO tested whether use of OPV or IPV would improve mucosal immunity. They conducted a randomised clinical trial in northern India in which almost 1,000 infants and children were given one vaccine or the other.
After four weeks, all the participants were given a “challenge” with the live-attenuated OPV to test whether they were protected against infection. In those who had received IPV, the amount of virus shed in stools after OPV was greatly reduced, meaning these children were less infectious to others. The IPV group also produced more antibodies, meaning these individuals are better protected against polio disease.
Reference: H. Jafari et al. ‘Efficacy of Inactivated Poliovirus Vaccine in India’ Science 22 August 2014: Vol. 345 no. 6199 pp. 922-925 DOI: 10.1126/science.1255006.
Article text (excluding photos or graphics) © Imperial College London.
Photos and graphics subject to third party copyright used with permission or © Imperial College London.
Reporter
Sam Wong
School of Professional Development