Quantitative phase microscopy (QPM) extends qualitative phase contrast microscopy to provide a map of the optical pathlength through a sample and so provides morphological information such as cell size. We wish to use QPM as a low phototoxicity, label-free means to segment and track cells in a multimodalmicroscopesystem that also provides rluorescence readouts. To this end, we are actively exploring the technique described as differential phase contrast microscopy (DPC)1 that can be implemented by acquiring 4 images with an appropriate semicircular aperture in the back focal plane of the condenser lens.
The figure below shows the schematic of a transmitted light DPC microscope that can be implemented with a programmable LCD mask located in the back focal plane of the condenser lens2. By sequentially setting the LCD (Electronic Assembly, EA DOGL128S-6) to present the four semicircular masks (A,B,C,D) and thereby acquiring 4 camera images (IA,IB,IC,ID), two phase gradient images can be calculated. In an alternative approach, the illumination system can be replaced by a programmable LED array in the Fourier space of the condenser lens3. A quantitative phase image can then be calculated from these phase gradient images by deconvolving these phase gradient images with the phase transfer function. Tian and Waller2 have developed and shared elegant DPC software to retrieve the quantitative phase images from such phase gradient images and we have used applied this to obtain the quantitative phase contrast image shown below.
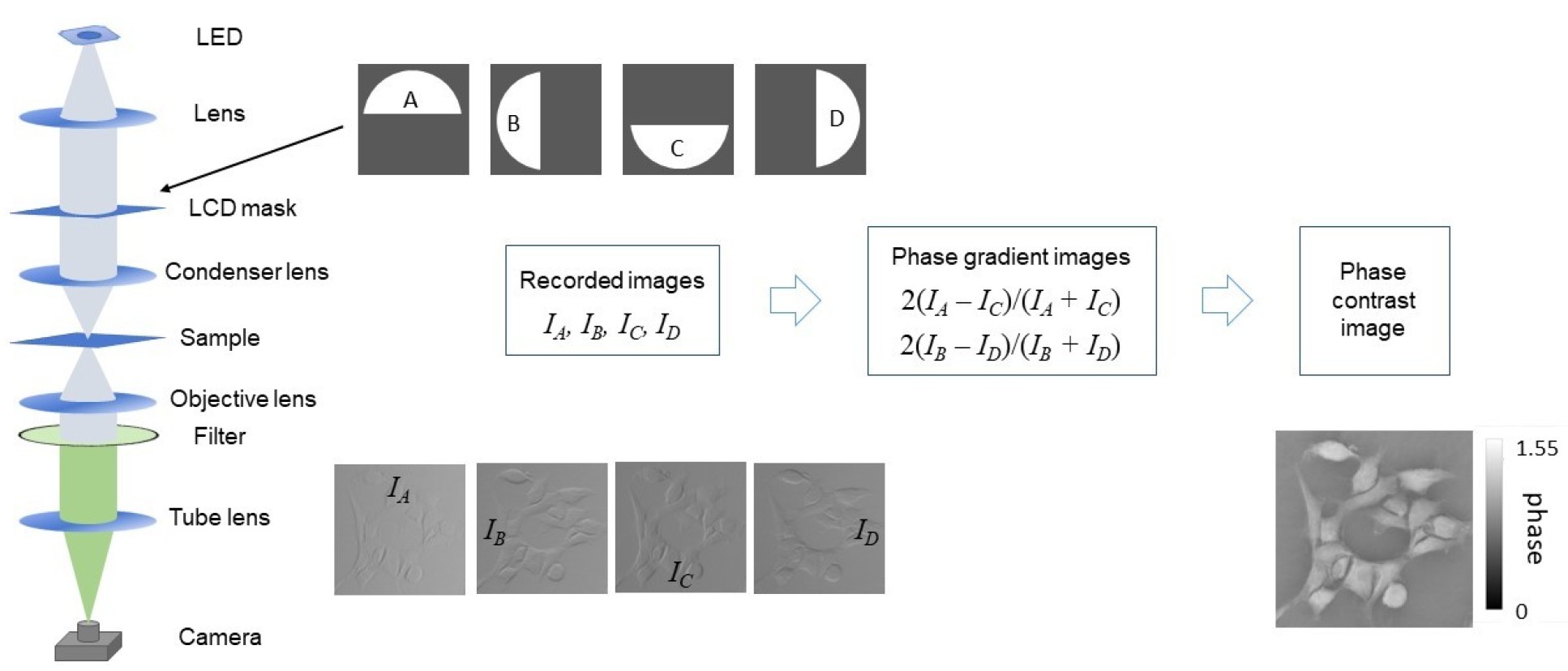
While this approach provides an inexpensive route to quantitative phase microscopy, it is limited for practical applications by the spectral response of the LCD, which provides maximum modulation at ~560 nm, and by the need to acquire 4 sequential images, which could lead to motion artefacts when imaging dynamic samples. Accordingly, we developed a new a single-shot approach to DPC micrscopy called polarisation phase differential phase contrast (pDPC) microscopy, which provides rapid semi-quantitaive phase images at up to 67 frames/second.
1 S. B. Mehta, C. J. R. Sheppard, Opt. Lett. 34 (2009) 1924
2 K. Guo, Z. Bian, S. Dong, P. Nanda, Y. M. Wang, G. Zheng, Biomed. Opt. Expr. 6 (2015) 574
3 L. Tian, L. Waller, Opt. Expr. 23 (2015) 11394