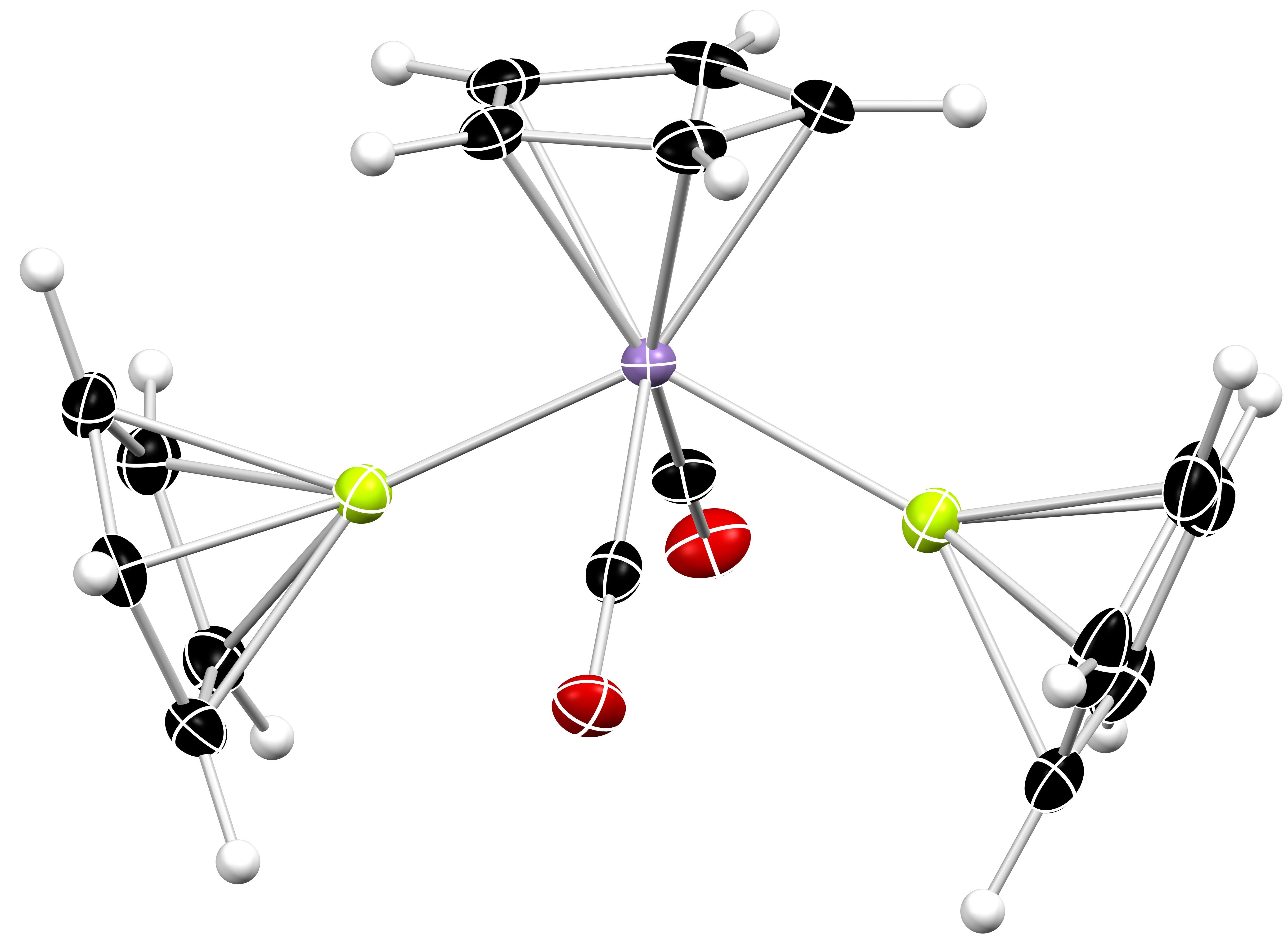
 Metal-Metal Bonding
Metal-Metal Bonding
Classical models of chemical bonding often fail to describe the true nature of an interaction between two (or more) metal atoms. A greater appreciation of the intricacies of metal-metal bonding is, therefore, an important fundamental pursuit. However, molecules featuring metal-metal bonds are also powerful tools; they can be considered soluble “molecular fragments” of bulk metal and can be conveniently probed by a plethora of analytical techniques that cannot be applied to solid materials. So, the targeted study of “exotic” metal-metal bonds can afford wide-ranging insights into the properties of some of the Periodic Table’s most unloved and unusual elements.
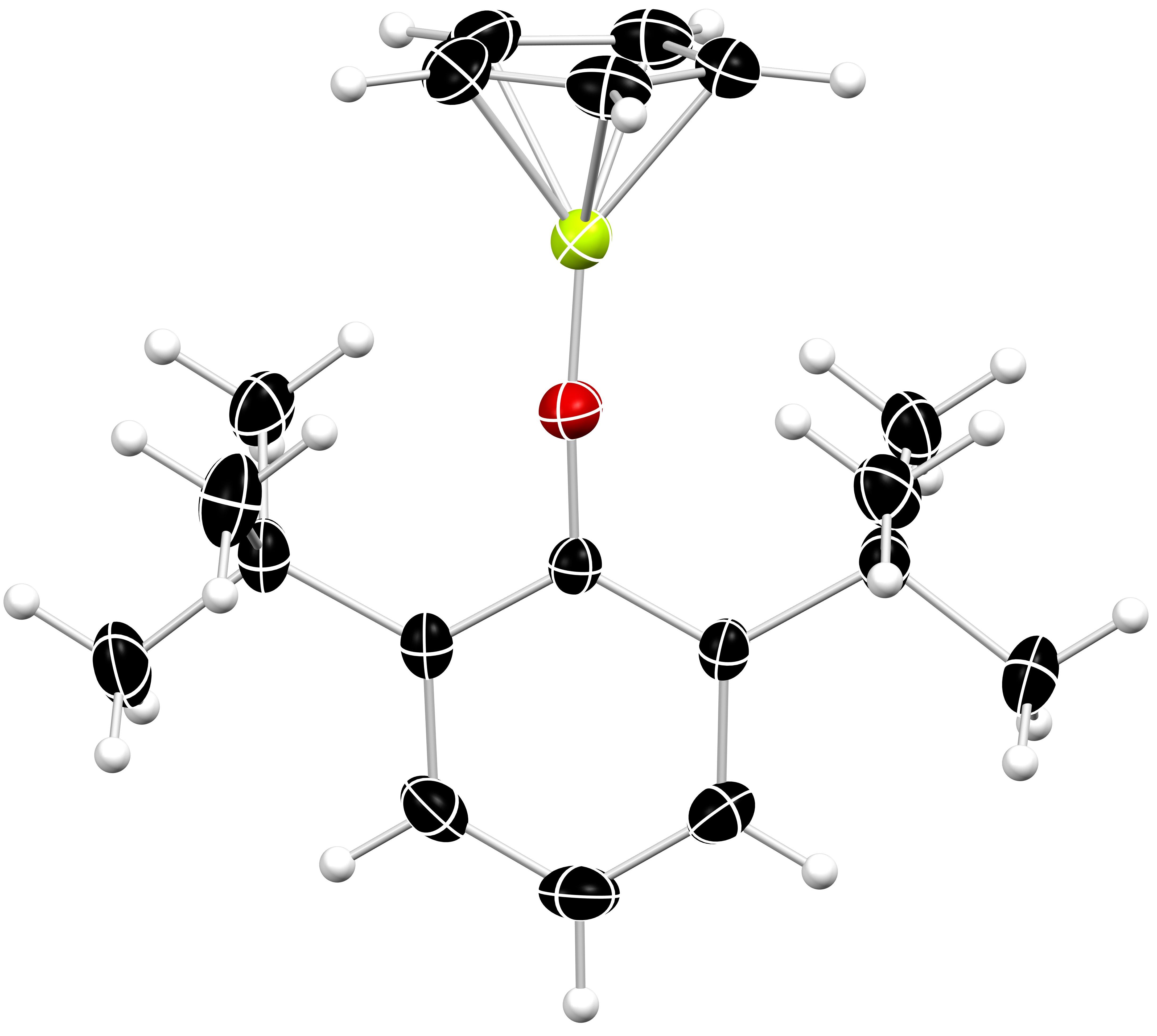 Alkaline Earth Metal Chemistry
Alkaline Earth Metal Chemistry
We are interested in the organometallic chemistry of the alkaline earth elements, with particular focus on beryllium and magnesium. Beryllium, the Periodic Table’s fourth element, is unique: its atoms are the smallest amongst all metals, it exhibits unsurpassed Lewis acidity, and its chemical bonds comprise a remarkably high degree of covalent character. Despite these intriguing properties, beryllium chemistry remains largely unexplored due to the element’s toxicity. Nonetheless, because the properties of the lightest elements underpin many of our bonding models, a comprehensive understanding of beryllium’s chemistry is of great fundamental importance.
Magnesium is one of the most abundant elements in the Earth’s crust. It is both inexpensive and non-toxic. These attributes make magnesium-containing complexes attractive candidates for application in challenging, industrially relevant chemical transformations. Additionally, magnesium organometallics − most notably Grignard reagents − are a cornerstone in both academic and industrial chemistry. Together, these factors provide strong motivation to deepen our understanding of this element.