Powered air-purifying respirators (PAPRs) help doctors and nurses treat coronavirus patients
Imperial College London researchers have developed and sourced powered air-purifying respirators (PAPRs) to assist medical staff to perform high-risk surgical operations. The high-quality PAPR respirators have been tested and used for risky surgical operations by NHS hospitals in London, including Imperial College NHS Hospitals, Guy's and St Thomas' Hospitals, London Lewisham hospital, and University College London Hospitals. We are now facilitating the supply of PAPR PPE equipment to assist UK surgeons and doctors to fight against the coronavirus and save lives.
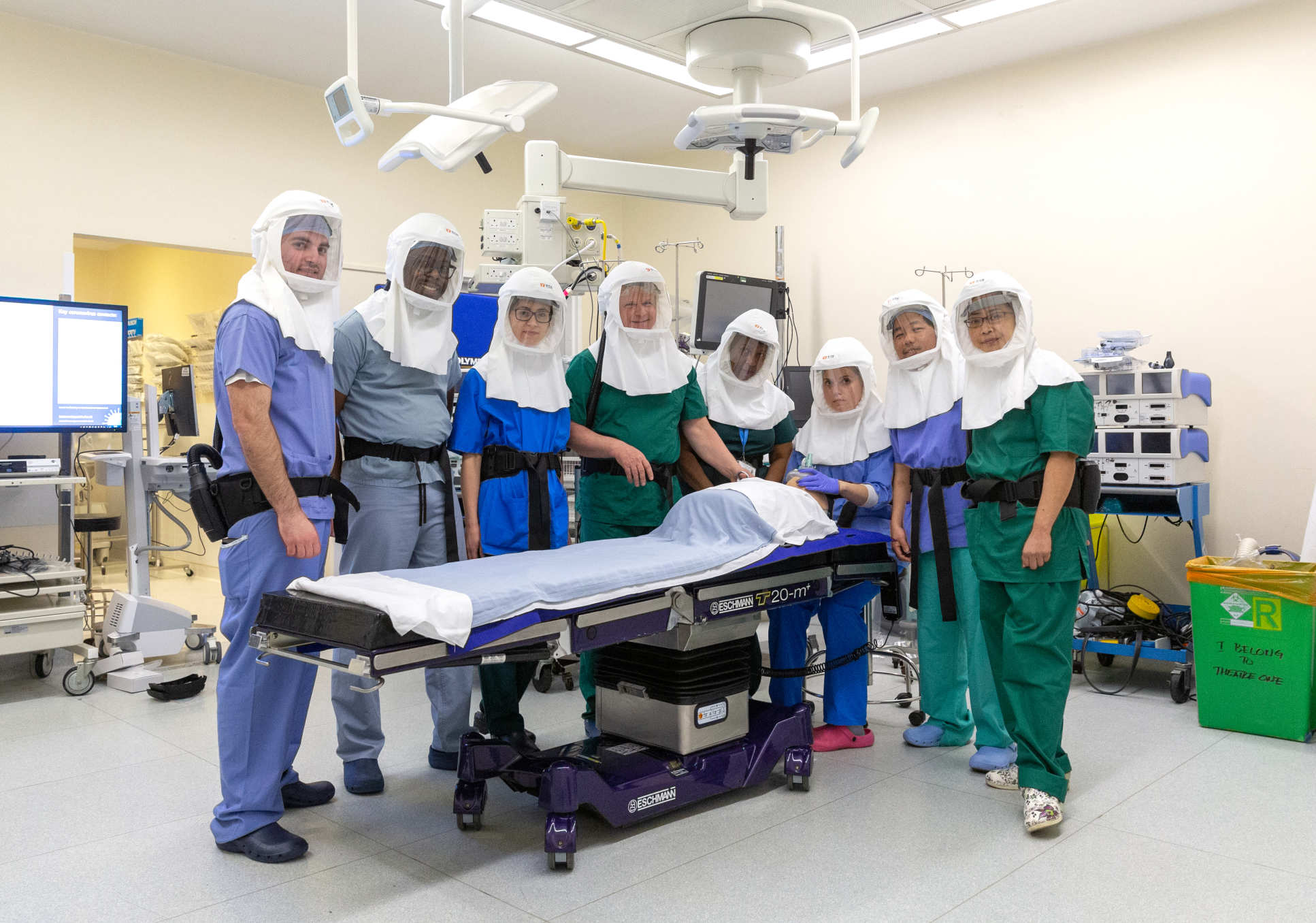
COVID-19 - Global Healthcare Challenge
Coronavirus represents the greatest global healthcare challenge for over a century. COVID infection has caused many patients to be admitted to intensive care as a consequence of viral pneumonia and respiratory distress syndrome. Patients have required intubation to provide the delivery of oxygen-rich air from ventilators.
As patients progressed through their illness several ultimately required a procedure known as Tracheostomy. This is where a tube is placed into the patient’s trachea or windpipe. Studies in Covid-19 infection have revealed that viral RNA can be detected in tracheal secretions many weeks following illness onset. Early reports during the pandemic also revealed a susceptibility to contracting infection among ENT surgeons. Chinese doctors and nurses have made an extraordinary contribution to defeating the coronavirus. Early experiences from Wuhan showed that infection in ITU workers was only resolved despite wearing N95 masks when PAPRs (Powered Air Purifying Respirator) became available.
Public Health England guidance for aerosol-generating procedures advises the use of an FFP3 mask. Surgeons have raised concerns about fit testing for a mask type only to be subsequently dispensed with a different brand on the day of surgery. There are also concerns for surgeons with facial hair for religious or cultural reasons (read the news; Telegraph report). Powered air-purifying respirators (PAPRs) provide full coverage for wearers with facial hair and work well as an alternative PPE that allows healthcare professionals to continue working during the pandemic while maintaining their unshorn hair (read news).
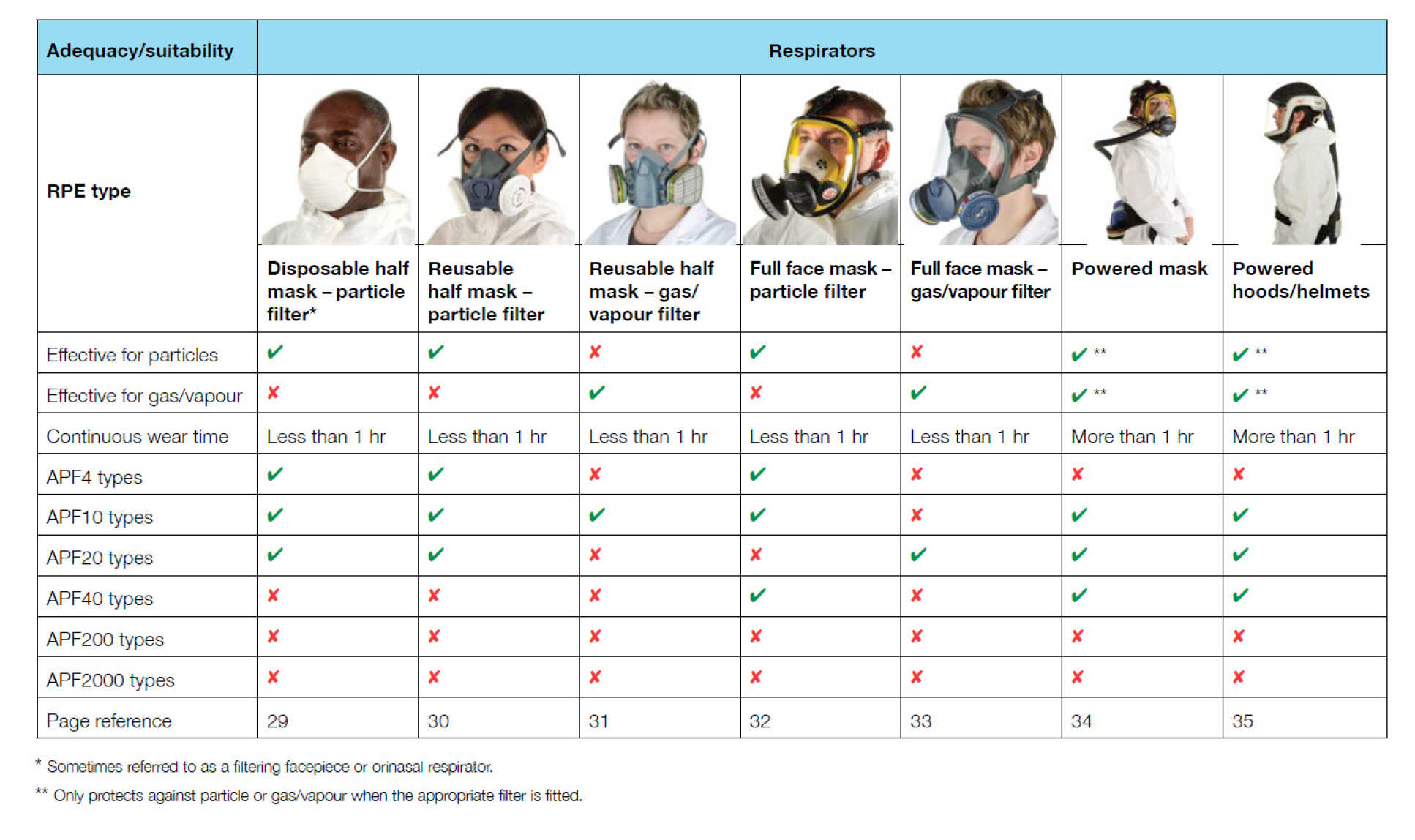
PAPR has been widely used and listed as an option by many international health organisations for PPE against coronavirus. UK Health & Safety Executive Law (HSG53 4th Ed 5.2013) provides detailed guidance on respiratory protective equipment at work (download the PDF). HSE law states that “FFP3 masks should not be worn for operations taking longer than 60 minutes, and lists various types of non-powered respirators and PAPRs. However, there is a significant shortage of PAPR PPE in the UK.
International Multidisciplinary Team
The project involves close collaboration between scientists and engineers at Imperial College, medical staff from UK and China, Imperial NHS trust, manufacturers and suppliers in China and the UK.
The project was initiated in mid of March and coordinated by Dr Qilei Song (Lecturer in Chemical Engineering), with support from Prof. Nilay Shah (Head of the Department of Chemical Engineering), and the Admin team of Department of Chemical Engineering at Imperial College. Professor Neil Tolley leads the team of surgeons in the Ear, Nose and Throat (ENT) division at Imperial College NHS hospitals.

An international team including Imperial academics, admin team, doctors of Imperial NHS Hospitals, Guy's and St Thomas' Hospital, London Lewisham hospital, University College London Hospitals, and support from Chinese doctors, UK and China industry partners. First Row: Dr Qilei Song, Prof. Nilay Shah, Prof. Neil Tolley, Anusha Sri-Pathmanathan, Dr Rongjun Chen, Prof. Daqing Ma, Prof. Kang Li, Prof. Paul Fennell; Second row: Viji Pilai, Sevgi Thomson, Nazma Mojid, Paul Silverster, Dr Asit Arora, Dr Tony Jacob, Prof. Jonathan Hughes, Third row: Prof. Peter Andrews, Prof. Chaozong Liu, Prof. Alister Hart, Prof. Johann Henckel, Prof. Haibo Qiu, Prof. Chun Pan, Prof. Jianfeng Xie, Prof. Yingzi Huang; Fourth row: Prof. Peiyong Li, Prof. Wei Chen, Prof. Xiang, Dr Shijiang Liu, Prof. Qingtao Meng. Industrial collaborators: Tony Wu, Nigel Nixon, and Steve Nixon.
Powered Air-purifying respirators (PAPRs) for COVID-19
A PAPR system is described as offering enhanced PPE. It consists of a battery-powered respirator that filters surrounding air by removing hazardous particles and contaminates via a HEPA filter (high-efficiency particulate air filter). This system consists of a protective hood that provides full protection of the head and coverage of the shoulder. It is extremely comfortable to wear due to the flow of positive-pressure clean and cool air over the face. The hood benefits from no uncomfortable tight-fitting seals on or around the face, making it particularly suitable for racial/ethnic/religious groups that cannot shave facial hair or fail the FFP3 mask fit test. A fit test is not required for wearing a loose-fitting PAPR hood.
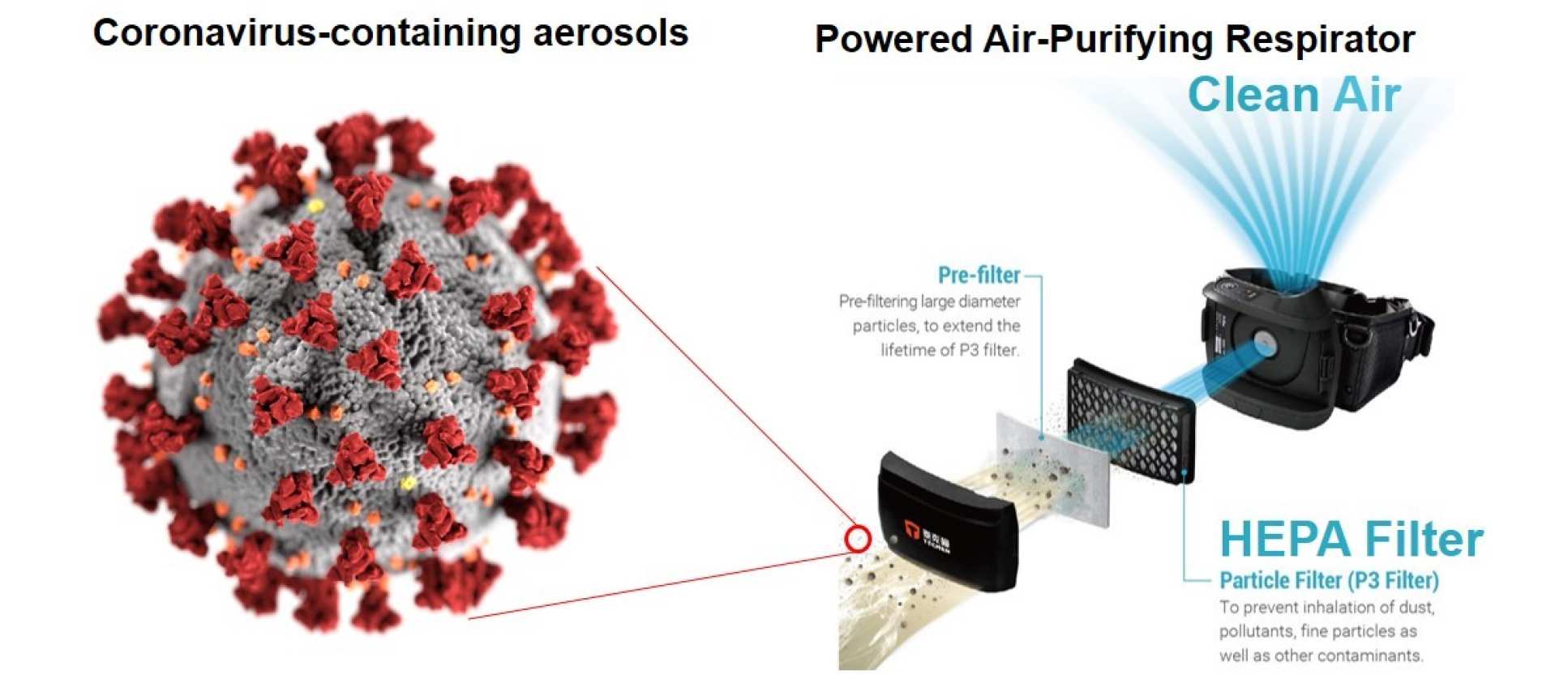
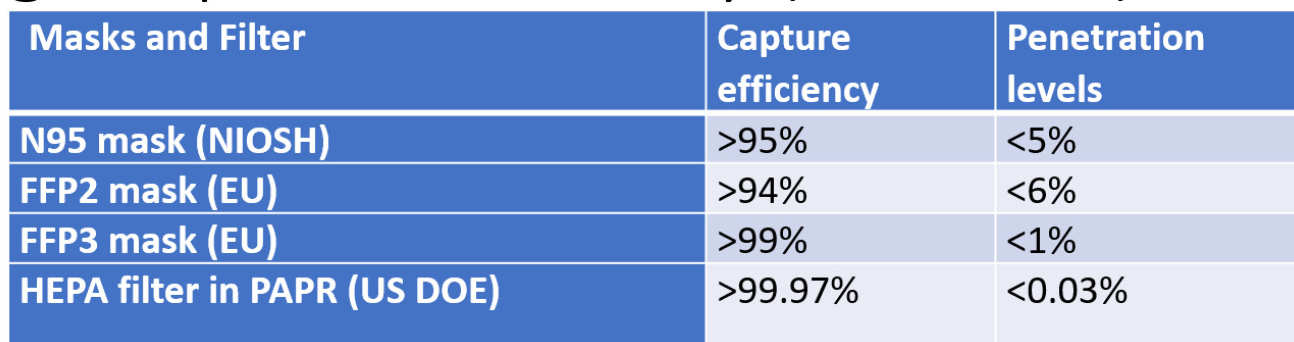
Comparison of capture efficiency of masks and HEPA filters
PPE type (UK Health & Safety Executive Law, HSG53 4th Ed 5.2013)
Link to the Respiratory protective equipment at work - A practical guide
Download the PDF copy of the guidance
Engineering Solution
Time is everything. We firstly evaluated the supply chain of PAPR and the availability of PAPRs in the UK. Chinese doctors have made tremendous efforts to defect the coronavirus. We knew that PAPRs have been widely used on the front line in Wuhan, such as Chinese-made TECMEN PAPR. In contrast, there is a significant shortage of PAPRs in the UK. Sourcing and supplying large volumes from China to the UK through the NHS Trust Procurement was challenging. Surgeons desperately need the PPE to perform surgeries, therefore we had to come out with an immediate solution. We found that PAPRs have been widely used in the UK welding industry. We immediately sourced several commercially available PAPRs and respirative hoods from a variety of UK suppliers and assembled a few prototypes in a week.
Safety is everything! PAPR consists of a variety of key components: air blower, prefilter, particulate filter, breathing hose, hood, and battery. Filter is the critical component. Many welding PAPRs are only equipped with P2-level filter that only gives nominal 94% capture efficiency. The filter can be contaminated by the coronavirus and needs to be changed frequently. The front covers in air blower units used in the welding industry have holes and are not intrinsically safe for use in hospitals. Contamination of fluids such as blood, water, alcohol and disinfectant can damage the filter and drastically reduce protection. The breathing tube and air blower should be easily cleaned and decontaminated.
After quick screening and evaluation of all these parameters, we chose the TECMEN PAPRs with P3-level high-efficiency particulate air (HEPA) filter from the UK distributor, Weldfast UK. We assembled a few TECMEN PAPR prototypes and tested them with Prof. Tolley's team.

Support from Chinese Manufacturers
We also contacted the manufacturer of TECMEN PAPR, Nanjing TECMEN Electronics in Nanjing China. TECMEN kindly donated their upgraded TECMEN PAPR TM-H2 systems and accessories, including front cover, and reusable hoods, and arranged speed delivery to Imperial College. Their kind donation allowed us to upgrade our prototypes to the same as TECMEN PAPR TM-H2 System that has been widely used in China.
We further purchased the TECMEN PAPR TM-H2 System from the manufacturer in China.
- TECMEN TM-H2 PAPR has been CE marked and certified to British Standard BS EN 12941:1998+A2:2008, by an independent PPE testing laboratory at INSPEC International Ltd, a UK Notified Body.
- The PAPR is certified to have high capture efficiency (>99.996%).
- The air blower unit is protected with a liquid-proof filter that helps to protect the filter from contaminations of fluids.
- The hood has a large clear visor that is anti-fog, so vision will always be clear.
- The battery duration is up to 9 hours with a recharge time of only 1 hour.
- The hoods are extremely comfortable to wear, and the turbo conveys filtered clean air to the head and face of the wearer.
- The manufacturer in China can supply key consumables (prefilter and filters, to be disposed on a daily basis for best protection) and accessories by air freight in 5-7 days.
- The manufacturer will provide detailed donning and doffing procedure, disinfection instructions, and share experiences from frontline doctors in China.
- Training procedure will be available from the ENT team at Imperial College Hospitals.
Therefore, these PAPR respiratory protective devices provide great support for medical staff to perform high-risk COVID-19 operations in a hospital environment.

Support UK NHS Hospitals
With the support of industry partners TECMEN and WeldFast UK, we have delivered and donated more than 100 PAPR respirators to the following NHS hospitals. We are also collaborating with these hospitals to perform research using this equipment.
- Imperial College NHS hospitals including Hammersmith Hospital, St Mary's Hospital, and Charing Cross Hospital
- University Hospital Lewisham, London
- Guy’s and St Thomas’ Hospital Affiliated to King’s College London
- University College London Hospitals including Royal National Throat, Nose and Ear Hospital
We hope to supply the equipment to many other NHS hospitals in UK. If you wish to support this project, please make a donation:
https://www.crowdfunder.co.uk/hoodsforheroes#start
https://www.justgiving.com/crowdfunding/ppe-for-frontline
Industrial Partners
We have established the supply chain by connecting the manufacturer in China, Nanjing TECMEN Electronics, and UK distributor WeldFast. The collaboration allows the UK hospitals to purchase large volumes at reasonable prices, at a time when raw materials are extremely scarce.
TECMEN is a reputable manufacturer located in Nanjing, Jiangsu Province, China. The company is certified to product quality management (ISO 9001/2000 certified). TECMEN brand products are CE, GS, DIN, CSA, ANSI, AS/NZS approved already under the newest standard.
Website: www.tecmen.com Email: tecmen@tecmen.cn
WeldFast UK is a leading UK company supplying engineering and safety equipment including PAPR. Weldfast UK currently supplies and supports over 800 distributors in the UK and Ireland with the emphasis on supplying quality equipment and services at competitive pricing.
Website: https://www.weldfastuk.co.uk/ Email: sales@weldfastuk.co.uk
Collaboration with Chinese Doctors
Chinese doctors and nurses made tremendous efforts to beat back the coronavirus on the front line in Wuhan. Dr Song and colleagues at Imperial, including Dr Rongjun Chen from the Department of Chemical Engineering and Professor Daqing Ma, from the Department of Surgery & Cancer, contacted frontline doctors in Wuhan, China who have been sharing their clinical experiences and knowledge with the hospital staff, as well as experiences of using this equipment. Many Chinese Students studying in the UK also volunteered to help collect feedback and translation of practices on COVID-19 treatment published by Chinese professional societies and journals.
An international zoom meeting with Imperial College NHS Healthcare Trust has taken place which provided a great deal of knowledge, insight, and advice to the doctors of Imperial hospitals. The Chinese doctors working day and night at Wuhan front line, including Professor Haibo Qiu and his team from Zhongda Hospital at Southeast University in Nanjing, Professor Xiang Lu and his team from Tongji Hospital at Huazhong University of Science and Technology in Wuhan, Dr Peiyong Li and Dr Wei Chen from Ruijin Hospital in Shanghai, Dr Shijiang Liu from Jiangsu Provincial People's Hospital of Nanjing Medical University in Nanjing, spoke with medics from Imperial College Healthcare NHS Trust to establish a step-by-step for safely using and re-using the equipment.
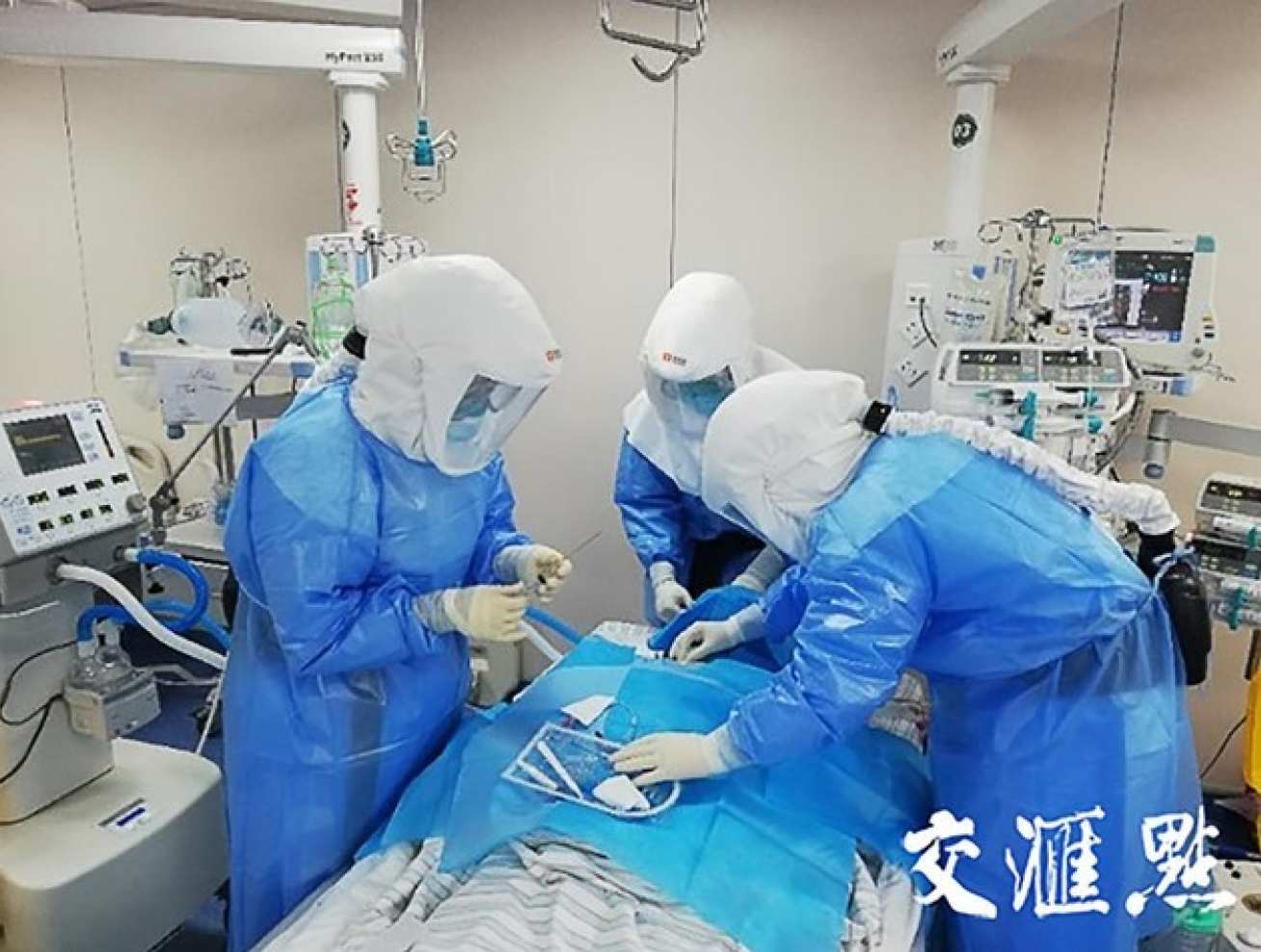
Academic Impact and Societal Impact
Global Tracheostomy collaborative Webinar
Prof. Neil Tolley has shared their early experiences of using PAPR with UK ENT association (Read the newsletter by Prof. Tolley) and the broader clinical communities in Europe and worldwide through a Global Tracheostomy collaborative Webinar - Overcoming COVID-19 through Science and Teamwork: Lessons from the United Kingdom, Italy, and United States.
Moderators:
Michael Brenner MD, FACS, Michigan Medicine | University of Michigan
Vinciya Pandian PhD, MBA, MSN, RN, ACNP-BC, FAANP, FAAN, Johns Hopkins University
Speakers:
Professor Neil S. Tolley MD, FRCS, DLO
Senior ENT-Thyroid Surgeon
Imperial College Healthcare NHS Trust, London, UK
Dr. Peter J. M. Openshaw, FRCP
FMedSci Immunologist and Member of the New and Emerging Respiratory Virus Threats Advisory Group (NERVTAG)
Imperial College Healthcare NHS Trust, London, UK
Professor Paolo Pelosi, MD, FERS
Director, Chief Professor of Anaesthesia and Intensive Care Director, Specialty School in Anaesthesiology
University of Genoa, Genoa, Italy
Head of the Anaesthesia and Intensive Care Unit at the IRCCS, San Martino-IST Hospital, Genoa, Italy
Dr. Christopher Rassekh, MD, FACS
Professor, Department of Otorhinolaryngology-Head & Neck Surgery Co-Chair, Hospital University of Pennsylvania Airway Safety Committee
Director of Risk Reduction and Director of Professional Practice, Department of OtorhinolaryngologyHead and Neck Surgery
University of Pennsylvania, USA
Dr. Asit Arora, PhD, DIC, DOHNS, FRCS
ENT, Airway, Robotic Head & Neck Consultant Surgeon
Guy’s and St. Thomas’ Foundation NHS Trust, London, UK

Publication on international consensus guideline document of tracheostomy
Prof. Neil Tolley and Dr Asit Arora are lead authors on an international consensus guideline document of tracheostomy published in the Lancet. Knowledge and visibility of the role of PAPRs have been shared with all UK ENT surgeons who have already performed hundreds of tracheostomies.
Link to the paper: https://www.thelancet.com/journals/lanres/article/PIIS2213-2600(20)30230-7/fulltext
Download the PDF copy of paper: https://www.thelancet.com/action/showPdf?pii=S2213-2600%2820%2930230-7

Useful Resources and Videos
Below you can find a range of videos available on YouTube showing the donning and doffing procedure of using PAPR. Note that these videos are provided only as references. Imperial College does not take any responsibility for the use of these videos. The infection control team in hospitals are recommended to develop their best practices and procedures according to their hospital facility and settings.
Donning and Doffing Process of TECMEN PAPR (Warning: not in a hospital environment)
TECMEN PAPR instruction manual
Donning N95 mask and PAPR
https://www.youtube.com/watch?v=NPGHgUs0Ptc
Donning PAPR
Doffing of PAPR
https://www.youtube.com/watch?v=yVnQiCrdTrQ
Doffing of PAPR for COVID-19
How to Safely Put on PPE, Selected Equipment: PAPR and Gown
https://www.cdc.gov/vhf/ebola/hcp/ppe-training/PAPRRespirator_Gown/donning_01.html
How to Safely Put on PPE, Selected Equipment: PAPR and Gown
How to Safely Take off PPE, Selected Equipment: PAPR and Gown
https://www.youtube.com/watch?v=-X4dNMgFGyo
How to Safely Take off PPE, Selected Equipment: PAPR and Gown
How to Safely Put on PPE, Selected Equipment: PAPR and Coverall
https://www.cdc.gov/vhf/ebola/hcp/ppe-training/PAPRRespirator_Coveralls/donning_01.html
How to Safely Put on PPE, Selected Equipment: PAPR and Coverall
How to Safely Take off PPE, Selected Equipment: PAPR and Coverall
How to Safely Take off PPE, Selected Equipment: PAPR and Coverall
COVID-19 PAPR with Head Cover Donning and Doffing Procedure - For Highest Risk
Donning and doffing procedure of PAPR PPE for the use by the provider with the highest risk during aerosol-generating procedure with COVID-19.
COVID-19 PAPR with Head Cover Donning and Doffing Procedure - For Highest Risk
Frequent Asked Questions
To update
Acknowledgment
Emily Yeung, Fisher Scientific
References:
Joshua K. Tay; Mark Li-Chung Khoo; Woei Shyang Loh. Surgical Considerations for Tracheostomy During the COVID-19 Pandemic - Lessons Learned From the Severe Acute Respiratory Syndrome Outbreak. JAMA Otolaryngol Head Neck Surg. 2020 Mar 31. doi: 10.1001/jamaoto.2020.0764.
Handbook of COVID-19 Prevention and Treatment. https://gmcc.alibabadoctor.com/prevention-manual
https://www.cdc.gov/vhf/ebola/hcp/ppe-training/PAPRRespirator_Gown/donning_01.html
https://www.cdc.gov/vhf/ebola/hcp/ppe-training/PAPRRespirator_Coveralls/donning_01.html
Letter of Prof. Neil Tolley to UK ENT Association - Powered air-purifying respirators (PAPRs) and COVID-19 https://www.entuk.org/powered-air-purifying-respirators-paprs-and-covid-19-0
Rapid Ramp-up of Powered Air-Purifying Respirator (PAPR) Training for Infection Prevention and Control during the COVID-19 Pandemic. Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7158777/
USA CDC guidance on PAPR equipment
Preparing for a COVID-19 pandemic: a review of operating room outbreak response measures in a large tertiary hospital in Singapore
https://link.springer.com/article/10.1007/s12630-020-01620-9
What Your Employers Should Provide: Protections at Work for COVID-19
"PPE for COVID-19 must include, at minimum, N95 respirators or higher, isolation gowns, eye protection, and gloves. Surgical and non-respirator face masks do not protect persons from airborne infectious diseases and cannot be relied upon for novel pathogens such as COVID-19. A Powered Air-Purifying Respirator (PAPR) with high efficiency particulate air filters must be worn during aerosol generating procedures on suspected or confirmed COVID-19 cases."
https://www.nationalnursesunited.org/california-nurses-know-your-rights-protections-work-covid-19-0
Contact Details
Department of Chemical Engineering
Imperial College London
South Kensington Campus
London SW7 2AZ
UK
Telphone Numbers:
Office: +44 (0)20 7594 5623
Email: q.song@imperial.ac.uk







