Increasing concerns related to the climate crisis and environmental pollution due to the excessive release of carbon dioxide have driven research efforts into green transportation. Electric vehicles, powered by lithium ion batteries are of particular interest. It is expected that 64% of the UK vehicle fleet will be electric by 2030 and that this will contribute to reducing air pollution and our dependency on dirty fossil fuels.1 In order to support this increased uptake in electric vehicles, current Li-ion battery technology needs to be improved in multiple key areas: performance, lifetime, and safety. To achieve such improvements, a comprehensive understanding of the degradation mechanisms of all components encountered during LIB operation needs to be developed in order to mitigate the processes—enabling safer operation and longer lifetimes.1
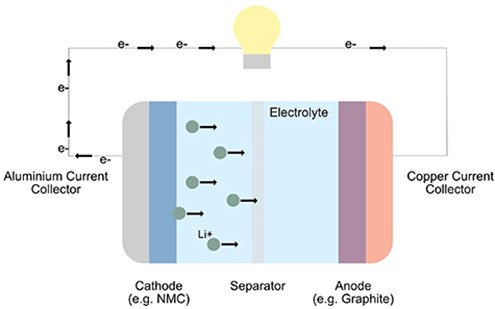
A Li-ion battery is a rechargeable, secondary battery. Its operation is based on the reversible intercalation of lithium ions into a crystal structure to store and release charge. A Li-ion battery cell is made up of a cathode and an anode, separated by a porous membrane, all wetted by the electrolyte.1
Our strategy
Many parasitic reactions in Li-ion batteries evolve volatile species as a side product so we have developed a new type of electrochemistry mass spectrometry with SpectroInlets to probe these reactions. With this real-time and picomole-sensitive technique, we are able to grow a fundamental understanding of the mechanisms that contribute to declining Li-ion batteries. We couple these measurements with complementary surface analysis studies to develop a holistic understanding of these complex mechanisms, enabling the development of mitigation strategies. We also look into alternative battery technologies such as Sodium (Na-ion) and Aluminium (Al-ion) batteries, interesting for their more abundant materials although in their earlier stages as compared to Li-ion battery technologies.
References
1. Thornton, Guo, Koronfel, Stephens, Journal of Physics: Energy 2021