Read and watch some examples of how animals are used in research at Imperial. You can find more examples on the Imperial News webpage.
Case studies
The amphibian plague
Around the world, frogs and other amphibians are facing rapid population decline and extinction – largely due to an aggressive fungal parasite. In this video Professor Matthew Fisher from Imperial’s School of Public Health explains how his work in the lab to help these animals is being successfully carried into the wild. His team is developing an antifungal treatment for frogs using a drug commonly used in humans. Finding in the lab the exact dose that they can use on an animal without harming it, they can then apply it in the wild and clear the infection in the wild population. The strategy was successful for the island of Majorca. The team will apply the system to more wild environments affected by different infections.
Ferrets and the flu virus
Each year millions of people are infected with flu and hundreds of thousands die as a result of the infection. In this video, Professor Wendy Barclay explains why researchers use ferrets to study the flu virus and the role ferrets are playing in preventing infections round the world. Ferrets are naturally susceptible to the same virus as humans. Therefore, they can be used to study human flu directly. Dr Barclay experiments focus on how the transmission of the virus happens using low doses of the flu virus. Therefore, the ferrets used do not experience severe sickness. All of the vaccines for flu rely on ferrets for their generations.
Mice sleeping patterns and anaesthesia
General anaesthesia is routinely used to enable patients to withstand medical procedures that would otherwise inflict unbearable pain and result in unpleasant memories. In a similar way to anaesthesia, sleep is also characterized by altered consciousness and greatly reduced responses to sensory input. Despite the fact that sleep is one of the most powerful physiological drivers of behaviour and it clearly provides an essential biological need (long-term total sleep deprivation can cause death in rodents faster than food deprivation), exactly how sleep is triggered, and how it affects the neurological pathways of the brain is still a mystery.
 Rodents have been the most widely used model organisms in biomedical research for many years and their use has proved invaluable to understand the complexity of how receptors and channels influence brain physiology and generate sleep, or respond to anaesthetics to produce unconsciousness. With their genetic, biological and behavioural characteristics resembling those of humans, mice represent a powerful model to study the brain circuits. Genetic engineering of mice allows testing specific hypotheses regarding the importance of particular molecular targets or the importance of particular brain circuitry.
Rodents have been the most widely used model organisms in biomedical research for many years and their use has proved invaluable to understand the complexity of how receptors and channels influence brain physiology and generate sleep, or respond to anaesthetics to produce unconsciousness. With their genetic, biological and behavioural characteristics resembling those of humans, mice represent a powerful model to study the brain circuits. Genetic engineering of mice allows testing specific hypotheses regarding the importance of particular molecular targets or the importance of particular brain circuitry.
Professor Nick Franks and Bill Wisden’s research at the Imperial College focuses on the relationship between general anaesthesia, sleep and sedation. In their studies the two scientists theorise that the sedative and hypnotic actions of general anaesthetics may be mediated through overlapping neuronal pathways that control natural sleep. To test their theory, they use both tadpoles and mice. Tadpoles are helpful to study the potencies of anaesthetics metabolised in mammals. However, to get close to the human condition, mice are the standard model for brain research. The mice used in Prof Franks and Prof Wisden’s research usually experience mild discomfort, with full recovery and no long-term effects. The results obtained from those animals could lead to better anaesthetic drugs with fewer side effects.
‘Anaesthesia and sleep are both commonplace states that involve a reversible loss of consciousness, but we don’t understand how either works: every year, 250 million patients worldwide are given anaesthetics; and every day, at some point in the 24-hour cycle, all humans and animals require sleep. Our research suggests that sedation and deep sleep are intimately connected at the circuit level. Understanding how they interlink is important for both fundamental neuroscience and medicine’, says Prof Franks.
By exploiting what is known at the molecular level, their research should provide insights into both how natural sleep is regulated, as well as how and where general anaesthetics act at the level of neuronal networks.
The zebrafish heart and the human heart
Zebrafish, tropical freshwater fish, share 70% of human genes and have several body parts similar to us. Thus, many type of disease in humans could theoretically be modelled in zebrafish. Moreover, zebrafish embryos and larvae are completely transparent: their skin and internal organs as clear as glass. This make possible to follow the impact of a genetic manipulation or pharmacological treatment using non-invasive techniques and minimising animal suffering.
At Imperial College London, Dr Thomas Brand’s group is interested in defining what causes cardiac arrhythmia and muscle dysfunction.
The muscular system is responsible for the movement of the human body. Specialized muscle types are responsible for an array of functions in our body. For example cardiac muscle is responsible for the beating of our heart, and smooth muscle tissue is responsible for diverse functions such as the transport of food through our digestive system or the maintenance of an appropriate blood pressure in the vasculature. Diseases affecting any of these muscle systems can have devastating effects on the life of an individual. In particular cardiac muscles pathologies can be responsible for morbidity and lethality, while loss of ambulation and difficulties to breath can be the consequence of a failure of the muscular system.
Dr Brand was one of the first scientists to discover the Popeye-domain-containing (POPDC) genes, an evolutionarily conserved class of proteins, while studying novel genes involved in heart development. 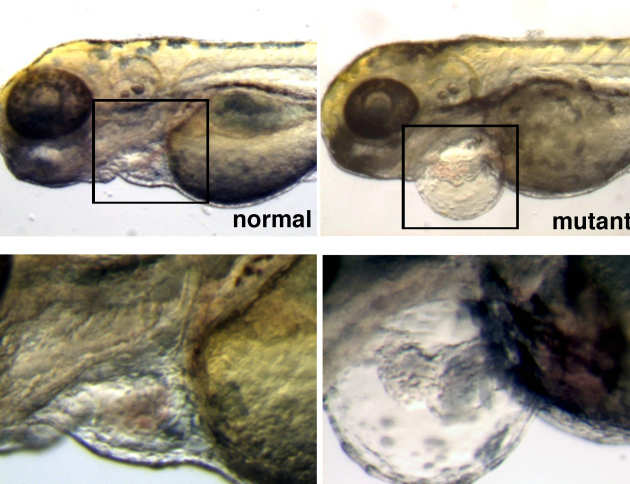
2) Dr Brand team use zebrafish and cell culture models to complement the efforts in the mouse. In particular, Dr Brand research have associated the Popeye genes with cardiac and muscle disease: expression of the POPDC disease-associated mutation in zebrafish caused similar phenotypes as in the patients displaying an overall reduction in heart rate and stroke volume and a gradual loss of proper function in the muscular system. Brand’s team makes all the efforts to ensure the well-being of the fish used for their experiments. Also in the most severe protocols, most fish recover quickly and behave normally.
There is no alternative to using animals for studying the complex inter-relationships between various cell types that orchestrate the molecular and cellular events that happen during cardiac injury. The zebrafish has recently emerged as a novel system to study cardiac electrophysiology and the development and physiology of the cardiac conduction system. In fact, despite significant anatomical differences, the electrophysiology and the beating rates of zebrafish and human hearts were found to be surprisingly similar.
The use of fish instead of mice also has the advantage of using a species with a lower capacity for experiencing pain. “Zebrafish is an ideal model to study some of the phenotypes in a shorter time frame and in a less invasive fashion and also for modelling human gene mutations”.
Thus, the zebrafish appears to be a powerful system to study the developmental and functional control of the cardiac conduction system.
